52 - 55
- Super User
- Category: Information
- 269
Appendix 1. Detailed descriptions of diver histories.
All audiograms for the cases are summarised in Appendix 2. The original copies of the audiograms have been withheld for privacy and anonymity.
Case 1
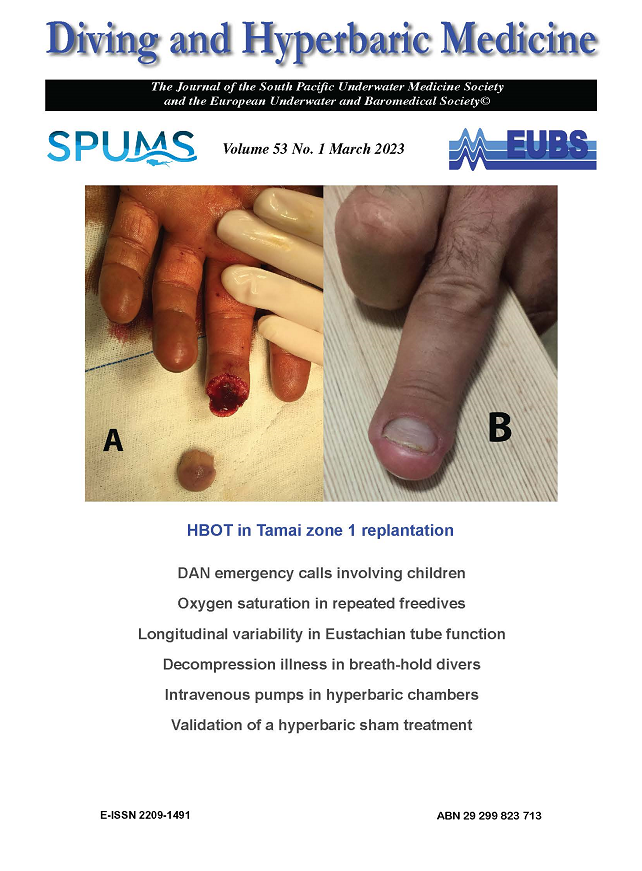
A 51 YO male scientific diver (Body Mass Index (BMI) = 26 kg/m2) with a normal audiogram at last medical. He was competing in an underwater hockey tournament and presented with hearing loss and tinnitus in the right ear (maximum depth 2.4 m). There were no vestibular symptoms. The subject had no previous issues or injuries to his ears from underwater hockey or diving, and all audiograms for his previous 20 years’ scientific diving medicals were normal. No compressed air diving had been undertaken in the week before or during the tournament. He was completely well before the tournament and denied any respiratory symptoms. Following the first day’s competition, he noted painless reduced hearing and tinnitus in his right ear. There were no ear clearing problems or other impact/injury. He played a further 6 games over 6 days with no change in his symptoms. After consulting a diving medicine specialist, an AC/BC audiogram was performed day 7 by an independent audiologist, before assessment day 8. Both tympanic membranes were normal appearance and the subject could demonstrate normal ear clearing. There were so symptoms or signs of vestibular disturbance, and full neurological examination was normal. The left ear was unaffected. The right ear audiogram was consistent with a mild to severe down-sloping sensorineural hearing loss affecting 1-8 kHz. No imaging was undertaken.
The diver’s case information was discussed with an Ear Nose and Throat (ENT) specialist, but was not reviewed in person by that specialist. In view of the likely causation from breath hold diving to 2.4m, a diagnosis of inner ear barotrauma was made. He agreed to a trial of HBOT following detailed instruction to undertake very gentle ear clearing. The subject was provided 9 treatments of HBOT, at 2.43 kPa (14:90:20 table, 3 x 5-minute air breaks). Prednisolone 50 mg daily was commenced day 2 after the second HBOT. No other specific treatment was advised apart from avoiding strong Valsalva manoeuvres and heavy lifting. Following two HBO treatments within 24 hours, and a single oral prednisolone dose, the subject’s hearing returned to normal range for 1kHz to 2kHz and had significantly improved in 3 and 4 kHz. He received ENT assessment with no changes to management. HBOT was continued daily to a total of 9 treatments. At no stage did he develop any vestibular symptoms. By end of 9 treatments, hearing in the right ear was normal for all frequencies except 6 kHz and 8 kHz. The subject received follow-up at 3, 6 and 12 months and his hearing has remained stable. After assessment by ENT and Diving Medicine Specialists, he was cleared for returning to occupational diving. He successfully resumed all diving activities including underwater hockey and has had no further issues with hearing.
Case 2
A 22 year-old male commercial diver (BMI = 24 kg/m2) had an uneventful 4-year diving career with more than 300 hours spent underwater. He received a comprehensive AS/NZS 2299.1 occupational diving medical one day prior to his incident, and did not dive that day. At that assessment, he demonstrated normal ear clearing, tympanometry and his audiogram was normal. He had no respiratory symptoms at the time of the medical and none on the day following. He undertook two brief dives the day after his medical, to 18 metres for 5 minutes each. The dives were separated by a surface interval under 15 minutes (a common practice for the industry). The diver noted no ear clearing difficulties, ascents were fully controlled and no symptoms were noted during or after the dive. He did not experience vertigo or disequilibrium at any stage, before, during or after the dives, and no lifting, straining, forced Valsalva, coughing or sneezing occurred. He was well when retiring to bed the same night. The next morning, he woke with hearing loss and tinnitus in his left ear. Of his own volition he returned to the same audiologist who had performed his AS/NZS 2299.1 audiogram, and asked for a repeat test. This demonstrated 60-85dB sensorineural hearing loss across all frequencies from 500 to 8 KHz. An urgent ENT/diving medicine assessment revealed normal tympanic membranes normal middle ear function and a negative fistula test, completely normal neurological examination, no nystagmus and sharpened Romberg test stable for 60 seconds. He agreed to a trial of HBOT following detailed instruction to undertake very gentle ear clearing. No imaging was undertaken before treatment. He was provided with a 14/90/10 hyperbaric oxygen treatment table the night of his presentation. Improvements in hearing were noted of 15-30dB across frequencies 500Hz-3KHz, immediately after the first treatment. It was decided to continue HBOT on a daily basis and Prednisolone 50mg was prescribed after the second HBO treatment. By day 10 (10th HBOT), hearing had improved in the left ear, with mild impairment only in 6-8kHz. High resolution CT scan of his petrous temporal bones was normal. He was reviewed by an ENT specialist, and no further treatment was advised. His hearing recovery persisted to 8 months at follow-up. After detailed discussion regarding his diving career, it was decided to advise against further diving. He is reconfiguring his career to train as a Ship’s Master.
Case 3
A 52-year-old recreational snorkeler (BMI not calculated, but not clinically obese) had a previous history of high frequency hearing loss from 3 – 8 kHz due to industrial noise. He was breath-hold diving for abalone and crayfish to a maximum of 4-5 metres depth. He undertook multiple descents and ascents over a 30-minute period. He experienced difficulty in clearing both ears and experienced pain and diminution of hearing in his right ear. He had no vertiginous symptoms. He denied respiratory symptoms prior to his breath-hold dives. He presented to his GP who referred him to an ENT specialist. After a normal MRI scan, he was prescribed 50mg prednisolone daily for 10 days. After no benefit from steroids, he was then referred for HBOT. His audiogram at the commencement of HBOT was identical to post injury 10 days before, demonstrating 55-70 dB sensorineural hearing loss across all frequencies right ear. Neurological examination was normal, with no signs of vestibular dysfunction. Middle ear clearing was normal at the time of diving specialist assessment. Given failure to improve with steroids, HBOT was commenced as a possible salvage. He was provided 10 daily HBOT using 14/90/20 or 14/90/10 schedules. There was normalisation of his hearing in 500-1.5 kHz and significant improvement of 2 kHz, following three hyperbaric oxygen treatments. He completed the 10 treatments and at discharge, was counselled against further breath-hold diving. His hearing remained stable at follow-up 6 months after treatment.
Case 4
A 31-year-old male commercial diver (BMI = 27 kg/m2) with 10 years’ experience, had a previous history of mild musculoskeletal decompression sickness, seven years prior to presentation. His previous annual dive medical health risk assessment showed normal hearing nine months before his injury. He had no symptoms or ear issues in the ensuing months. Just before his presentation, he been diving daily within dive table limits over a four-day period. There was no strenuous activity, no trauma, no ear clearing problems during his diving, and no respiratory symptoms. On the fourth day, starting with repetitive factor 1.0, he completed a single dive (with five-minute safety stop) to 15m for 53 minutes – he had no ear clearing issues and was symptom free immediately post dive. The evening after his dive, he noted reduced hearing in his right ear, “like water in the ear”, but did not report his symptoms until three days later (this period spanned a weekend). After reporting, his supervisors referred him for medical assessment. He had no symptoms or signs of vestibular dysfunction, no nystagmus, normal balance, normal tympanic membranes which cleared easily with gentle Valsalva, negative fistula test. His audiometry at day four showed sensorineural hearing loss in his right ear at four days post-diving across 500 Hz – 8 kHz. Impedance audiometry showed normal middle ear pressure with increased tympanic compliance. Findings were consistent with sensorineural hearing loss right ear, probably due to IEBT, however isolated cochlear decompression illness (although unlikely) could not be absolutely ruled out. Following discussion with ENT specialists, he was treated with a series of 14/90/20 tables for 10 consecutive days. Steroids were not prescribed. Audiometry was independently undertaken at the hospital’s audiology service, returned to normal after HBO treatment four. His hearing has remained stable and normal to 3 months follow up.
He received an MRI scan of his brain which demonstrated no abnormalities in his cranial nerves 7 and 8, and membranous labyrinth, and an incidental tiny cyst in the right sella turcica. High resolution CT scan was normal except for a slightly narrow right cochlear aqueduct (of uncertain significance). Following review and discussions with his employer, he chose not to dive again.
Case 5
A 62-year-old male hyperbaric professional (BMI = 27), with 3,500 hours recreational diving experience and no previous ear issues or barotrauma. Previous audiograms for occupational medicals had been normal with all frequencies demonstrating 20 dB or less hearing loss. Two uneventful dives were completed day one, well inside no-decompression limits, to maximum 21 m depth. The diver noted mild upper respiratory symptoms at the start of day 2. Following a 21-hour surface interval (at 0926), an uneventful multilevel dive was undertaken day two to 16 metres for 64 minutes, average 11.3 metres including five minutes at 3-5 metres safety stop. The diver had no problems clearing his ears on the first dive, day two. After a one-hour surface interval a second dive was commenced to 16 metres. The diver made camera adjustments whilst descending and omitted ear clearing. He noted pain in his right ear, which settled after some sub-surface ascents and further descent. The dive was completed with no further issues and no reverse ear squeeze, 57 minutes dive including a five-minute safety stop at 3-5 m. During the afternoon, the diver snorkelled with a number of free dive descents, and noted right ear stickiness when clearing. He noted a minor hearing impairment and crackling that evening. The diver woke the next day with significant hearing impairment and tinnitus in the right ear, and 2/10 pain. He was unable to hear conversation with the right ear when his left ear was occluded. He did not dive or snorkel again and flew home to Australia day 10 without ear clearing difficulties, although with some perception of moist crackling in the middle ear. Following air and bone conduction audiograms, he was assessed by an ENT specialist who diagnosed inner ear barotrauma with moderate sensorineural hearing loss. At the diver’s request, he was referred for HBO treatment, and received 5 daily treatments using 14:90:10 tables, and had no difficulties in clearing his ears. The diver chose to stop HBOT at treatment number 5, because hearing in the right ear had returned during treatment 2. Steroids were not prescribed. No imaging or further investigations were performed. He returned to diving one month later and has completed 65 more dives in 2023-24 without incident.
Appendix 2. Subject audiometry results detailed.
- Super User
- Category: Information
- 142
Applications should include a brief synopsis (1-2 pages) of the project and be Hazardous marine environments as defined in AS/NZS 2815.6 (2013).36
- Risk of entanglement – diving in and around nets and cages, multiple ropes and lines, tree branches, man-made underwater structures such as shipwrecks, sunken vehicles, or other sunken material.
- Diving in an overhead environment, without vertical access to the surface.
- Highly limited or zero visibility.
- Work near outflow or inflow to pipes.
- Diving in currents or fast flowing creeks, rivers and drains.
- Diving associated with setting of weights or moorings or use of lifting devices for anything other than fish or shellfish.
- Work in high boat traffic areas such as navigation channels, entries to marinas, operational ports.
- Super User
- Category: Information
- 203
Sokolowski et al., Supplementary File 1
Sokolowski et al., Supplementary File 2
Ovid MEDLINE(R) <1946 to June Week 4 2023> Supplementary File 2
1 exp Hyperbaric Oxygenation/ 12679
2 hyperbaric oxygen*.mp. 14339
3 exp Myopia/ or myopi*.mp. 27633
4 exp Cataract/ or cataract*.mp. 70354
5 vision chang*.mp. 382
6 exp Visual Acuity/ 92476
7 visual chang*.mp. 1015
8 diving.mp. or exp Diving/ 10028
9 (dive or diver or divers).mp. [mp=title, book title, abstract, original title, name of substance word, subject heading word, floating sub-heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms, population supplementary concept word, anatomy supplementary concept word] 7871
10 1 or 2 14339
11 3 or 4 or 5 or 6 or 7 167046
12 8 or 9 13736
13 10 and 11 and 12 4
14 exp Hyperoxia/ or Hyperox*.mp. 14445
15 10 or 14 28042
16 11 and 12 and 15 7
17 1 or 2 or 14 28042
18 11 and 17 202
19 (oxygen* toxic* or oxygen* poison*).mp. [mp=title, book title, abstract, original title, name of substance word, subject heading word, floating sub-heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms, population supplementary concept word, anatomy supplementary concept word] 1818
20 1 or 2 or 8 or 9 or 14 or 19 41790
21 11 and 20 248
22 (Nuclear cataract and myopia during hyperbaric oxygen therapy).m_titl. 1
23 "Hyperbaric oxygen therapy and the possibility of ocular complications or contraindications".m_titl. 1
24 "Side effects of hyperbaric oxygen therapy".fc_titl. and "2014".fc_pubyr. 1
25 "Hyperoxic myopia.".fc_titl. and "1978".fc_pubyr. 1
26 "Rates of visual acuity change in patients receiving hyperbaric oxygen in monoplace and multiplace chambers".fc_titl. 1
27 "Myopic shift and lens turbidity following hyperbaric oxygen therapy".fc_titl. 1
28 "Myopic Shift during Hyperbaric Oxygenation Attributed to Lens Index Changes".fc_titl. 1
29 "Short-term effects on ocular variables immediately after hyperbaric oxygen exposures".fc_titl. 1
30 "Ophthalmic side effects of hyperbaric oxygen therapy".fc_titl. 1
31 "Ocular refractive changes in patients receiving hyperbaric oxygen administered by oronasal mask or hood".fc_titl. 1
32 "Posterior segment changes of the eye during hyperbaric oxygen therapy".fc_titl. 1
33 "Hypermetropia-succeeded myopia after hyperbaric oxygen therapy".fc_titl. 1
34 "The myopic shift associated with hyperbaric oxygen administration is reduced when using a ".fc_titl. 1
35 "Ocular alterations in divers".fc_titl. 0
36 "Hyperoxic myopia in a closed-circuit mixed-gas scuba diver".fc_titl. 1
37 "Hyperoxic myopia: a case series of four divers".fc_titl. 1
38 "Oxygen exposure and toxicity in recreational technical divers".fc_titl. 1
39 "Pulmonary effects of submerged oxygen breathing in resting divers".fc_titl. 1
40 22 or 23 or 24 or 25 or 26 or 27 or 28 or 29 or 30 or 31 or 32 or 33 or 34 or 35 or 36 or 37 or 38 or 39 17
41 21 and 40 17
Sokolowski et al., Supplementary File 3
|
Reference number |
Author |
Study Design |
Scuba/ HBOT |
Population size |
Population |
Exposure |
Measurements |
Results |
Represented in graph |
GRADE |
|||
|
Number of treatments/dives |
Environment |
Myopic Shift |
Cataracts |
Reversibility |
|||||||||
|
13 |
Marín-Martínez |
Case Report |
SCUBA |
2 |
Case 1: 50 yo male, no medical or ophtalmological history. No glasses. |
Unclear |
Both were diving with high ppO2 and HBOT after each dive, depths and treatment plans not mentioned. |
Refraction, biometry, axial length, keratometry. |
Case 1: refraction |
Both cases had cataracts in both eyes |
Not mentioned. |
NO |
Very Low |
|
14 |
Butler |
Case Report |
SCUBA |
1 |
48 yo white male, CCR mixed gas scuba diver. No eye history, good health, no medication. |
21 days of diving with typically 3 dives per day. |
Average depth 21.3 msw, mean daily dive time 4.04 h. Mixed gases. Total of 84.8 h accumulated at 130 kPa inspired O2. |
Diver was examined by optometrist on 20th October 1997 and again after his trip on 18th of February 1998. |
Pre-dive measurements were -0.5 (R) and 0 (L) and post-dive -2 (R) and -1.5 (L). Thus, both eyes had a myopic shift of 1.5 D |
No cataracts. |
Refraction reversed towards hyperopia. At follow up 7 weeks later, the refraction measured was 0 (R) and +0.25 (L) |
YES |
Very Low |
|
16 |
Brügger |
Case Series |
SCUBA |
4 |
Four military divers with accounts suggestive of hyperoxic myopia. These divers have participated in research protocols at NEDU. |
5 consecutive days of 6 hour dives with an 18-hour surface interval. |
Dives in a pool at 3.7 m. 6-hour dives were evenly divided into two separated by a 5–10 min surface interval for hydration and food. Total exposure 40.5 hours at 101 kPa of 100% O2. |
Visual acuity measured with Snellen Chart. |
Divers complaint was in line with objective finding of at least one line worse on the Snellen eye chart in at least one eye. An estimated 0.25−0.50 D change on the day of reported blurred vision, which occurred between the last day of exposure and four days after completion of exposure. |
Not reported. |
All divers had spontaneous recovery of visual acuity 7−30 days after onset. |
NO |
Low |
|
25 |
Fock |
Cohort Study |
SCUBA |
14 |
No known pre-existing lung or ocular disease. 14 male CCR divers (mean age 46, BMI 27) provided 20 data sets. |
The total number of dives was unclear. The data was collected over three multiday expeditions. Diving was done twice a day, with a surface interval of approximately 4 hours. |
Mean duration of dives was 112 min. PO2 was maintained between 130−140 kPa. In some cases decompression was accelerated by increasing pO2 to 150−160 kPa or surface supplied oxygen used at 6 msw. |
Pre- and post-expedition as well as at regular intervals during the expedition by Snellen eye chart. |
Subjective difficulty with distance acuity was reported by the end of the expeditions in 18 of the 20 data sets measured. After 13 days the mean change in refraction was 0.4 D |
Not reported. |
One diver sought formal evaluation with an ophtalmologist. Change was 0.75 (L) and 1.0 (R) towards myopia. This resolved in 8 weeks. The recovery of the rest of the divers was not known. |
YES |
Low |
|
10 |
Gesell |
Case Report |
HBOT |
1 |
49 yo female with sacral osteomyelitis. Medical history for hypertension, asthma, and scleroderma. Other causes of cataracts excluded. |
11 weeks, 48 treatments in total. |
250 kPa for 90 min. |
Formal refraction and biomicroscopy with lens evaluation. |
Eyesight changed from -1.00 D (R) and -0.75 D (L) to -2.5 D in both eyes. |
Cataracts stabilized at grade 1−2 and showed no evidence of reversal. |
No reverse. At 11 months both eyes at -4.25 D. |
YES |
Very Low |
|
11 |
Hagan |
Case Report |
HBOT |
1 |
58 yo female with thymic carcinoma, sarcoma, post-radiation lymphedema and recurrent chronic leg ulcers, elevated cholesterol, dilated cardiomyopathy, and controlled hypertension. Other causes of cataracts exlcuded. |
46 treatments. |
90 min, 200 kPa. |
Visual aquity and refraction was measured. |
Refraction change in both eyes |
Extremely large, four quadrant nuclear and cortical cataracts. |
Not reported. |
YES |
Very Low |
|
12 |
Evanger |
Case Report |
HBOT |
1 |
58-yo man treated for gingival cancer. |
21 days of HBOT. |
240 kPa for 90 min by oronasal mask. |
One week before HBOT routine eye examination and after two weeks of completion. Refractive state assessed with autorefractor on each eye. Monocular acuity (Snellen chart). |
After first series of HBOT myopic shift of -1.37 D |
Not reported. |
After the first series there was some reversal at 8 weeks. After second series, at 11 week visit a hypermetropic shift occurred + 1.62 D (R) and +1.50 D (L), which remained stable at 78 weeks (1.5 years). |
YES |
Very Low |
|
15 |
Fledelius |
Case Series |
HBOT |
17 |
12 men and 5 women, age range 24−72 (median 58) with post radiation osteonecrosis of the mandible. Patients with cataracts were excluded. |
30 treatments, 6 weeks 5 days a week via mask. |
95 min of 95% O2 at 250 kPa. |
Visual acuity |
Median change of refraction |
No cataracts. |
All patients undergoing post-treatment examinations returned to pre-treatment status. |
YES |
Low |
|
17 |
Anderson |
Cohort Study |
HBOT |
10 |
Mean age 59 (51−69) treated for osteoradionecrosis |
40 consecutive treatments, |
200 kPa for 120 mins at 95% O2. Total of 80 hours. External eye exposed to oxygen. |
The Snellen visual acuity, refractive error, applanation intraocular tension, and corneal curvature were measured within three days of the first exposure, and again at the end of the treatment. |
During the period of hyperoxic treatment there was an average change in myopia of about 0.25 D per week. |
Not reported. |
At the last follow-up (12.5 weeks in average) examination, the refractive error differed from the pretreatment value by -0.23 D. |
YES |
Low |
|
18 |
Palmquist |
Cohort Study |
HBOT |
25 |
Mean age 65.5 years, persistent leg ulcers. Five of the patients were diabetic. |
The total number of treatments varied from 150 to 850 exposures during 2 to 19 months (one exposure= one hour). Daily treatments were given 7 days a week. |
Pure oxygen was administrated twice for 1 hours at 2−2.5 times normal atmospheric pressure (101 kPa). |
Eyes were examined after each 100 treatments and the more affected eye was selected. The corrected visual acuity tested by Monoyer's chart and the refraction by Donders's subjective method. The central fundus was also inspected. |
24/25 of the patients had a refractive change of >1 D in direction of myopia. Mean value of maximal change in refraction was -3.0 D. Maximum change occurred between 100−300 treatment hours in 24 patients. |
15/25 had clear lense at the beginning, 7/25 developed cataracts during treatment. |
In most cases myopia started to reverse during therapy, 11 patients had persistent myopia after six months of termination of HBOT. |
YES |
Low |
|
19 |
Churchill |
Cohort Study |
HBOT |
85 |
62% male, patients mean age 57. Treated for radiation injury, osteomyelitis, compromised flap, diabetic wound and other approved indications. Mean sessions 35. 34 in monoplace, 34 in multiplace and 17 in both. |
The number of treatments varied between 10−79, with the mean of 35. |
Monoplace: 200 kPa for 90 min (chamber filled with oxygen at 240−250 lmin-1) |
Snellen eye chart was used to monitor vision changes. If patient could read 50% or more correctly of the line they were given credit for it. |
32% had one line change and 28% had at least a 2-line worsening of visual acuity. Change rate was similar with monoplace and multiplace patients, but monoplace reached more frequently 20/40 or worse. Patients who used mask had no vision change. |
Not reported. |
Not reported. |
NO |
Low |
|
20 |
Riedl |
Cohort Study |
HBOT |
29 |
18−74 years (mean 61.7 years), no cataract surgery, no previous HBOT, prescribed less than 40 treatments, multichamber, oronasal mask. |
40 treatments, Monday to Friday, standard regimen. |
Standard regimen was assumed to be 90 min at 240 kPa of 100% O2. |
Minor eye examination every 10 treatments and at 12 week follow up. In addition to ocular refraction and visual acuity, numerous other measurements were made. Cataract was graded using Lens Opacities Classification system. |
A myopic shift happened in 45 single eyes (77.6%) in 26 patients. Median change was |
A significant increase in nuclear colour and nuclear opalescence after the HBOT treatments, measured using the Lens Opacities Classification system. |
At 12 weeks follow up the change had normalized, but a small change (-0.25 D remained in the left eye). |
YES |
Low |
|
21 |
Evanger |
Cohort Study |
HBOT |
20 |
10 men and 10 women, with a mean age of 56.0 years, treated for radiation-induced osteonecrosis, proctitis or cystitis, all white. Inclusion criteria age below 70, normal ocular health, spectacle prescription within 5 D. No visual complaints. |
According to standard protocol. 20 treatments, 5 days a week, once per day. |
Monoplace hyperbaric chamber, 240 kPa over a period of 10−15 min, 90 min at the pressure. Three cycles of 30 min, interrupted by breathing compressed air by oronasal mask for 5 min between cycles. Decompression in 7−10 min. |
Objective refraction, measurements of forward scattered light, intraocular pressure, axial length of the eye, Scheimpflug picture, backwards scattered light. Baseline measurements were done after first HBOT session. Follow up measurement after 19 completed HBOT. |
All patients (n = 20) developed a myopic shift during treatment, |
No cataracts. |
Not reported. |
YES |
Low |
|
22 |
Evanger |
Cohort Study |
HBOT |
23 |
14 males, 9 females with average of 59. Subjects with healthy eyes, who were referred to six weeks of HBOT. |
HBOT Monday to Friday for 6 weeks. |
Monoplace chamber and compressed to 240 kPa within 10−15 min. 100% oxygen given for 90 min, interrupted every 30 min for 5 min of breathing air. Decompression 7−10 min. |
Objective measurements of refraction, corneal thickness, keratometry and intraocular pressure (IOP). Measurements done always before and after treatment |
Myopic shift developed at |
Not reported. |
Myopic shift reversion after weekends was noticed. No follow up after HBOT was over. |
YES |
Low |
|
23 |
Evanger |
Cohort Study |
HBOT |
32 |
Mean age 55.1 years. 20 patients by oronasal mask and 12 patients by a hood. Indications radiation induced osteonecrosis, proctitis or cystitis. Vision within +/- 5 D, normal anterior segment of the eye, Healthy eyes, no contact lenses allowed. |
HBOT for 21 consecutive days. |
Compression with air to 240 kPa within 10−15 min, 3x30 min 100% oxygen with periods of 5 min of air in between. Decompression to normal atmospheric pressure (101 kPa) in 7−10min. Multiplace chamber. |
Eye measurements were done a week before HBOT and 2–4 days after. They consisted of visual acuity, measurement of the power of corrective lenses for patients wearing distance spectacles and a slit-lamp examination of the anterior segment. Oronasal mask group had follow ups until 36 weeks and underwent additional examinations. Hood group had follow-ups until 10 weeks. |
A significant myopic shift ( > 0.5 D) was found in 24 (60%) single eyes in mask group and in 20 (83%) single eyes of hood group. Mean maximal change in mask group was -0.55 D (R) and -0.53 D (L). For hood patients the mean maximum change was -1.06 D (R) and -1.10 D (L). Myopic shift was larger in in patients given the hood. |
Not reported. |
For the mask group the myopic shift reversed in 6 weeks after HBOT treatment and remained unchanged for the rest of the follow up. |
YES |
Low |
|
24 |
Evanger |
Cohort Study |
HBOT |
15 |
Normal ocular health, ametropia within 5 D, < 70 yo, mean age 55.8 years. |
HBOT for 20 days from Monday to Friday. |
Treated in monoplace hyperbaric chamber. Compression with air to 240 kPa within 10−15 min, 3x30min 100% O2 with periods of 5 min of air in between. Decompression to normal atmospheric pressure (101 kPa) in 7−10 min. |
Baseline measurements included refractive state, axial length of the eye central retinal thickness, pulsatile ocular blood flow and intraocular pressure. The measurements were done after treatment on the first day of HBOT and the follow up after 19 days 2−7 hours after completion of HBOT. |
Significant myopic shift |
Not reported. |
Not reported. |
YES |
Low |
|
26 |
Bennett |
Randomized Controlled Trial |
HBOT |
120 |
Any patient that had been the given HBOT. |
20−30 HBOT sessions once per day, Monday to Friday for 4 or 6 weeks. |
240 kPa for 90 min. |
Visual acuity measured with Snellen Chart and investigators formally assessed subjective refraction. |
Myopic shift statistically greater in those using a hood (after 20 treatments mean change -0.92 D with hood vs. -0.52 D with mask). After 30 treatments change of |
Not reported. |
Patients follow up at 4 and 12 weeks. At 4 weeks hood group improved to a mean shift of |
YES |
High |
Sokolowski et al., Supplementary File 4. Table of the extracted oxygen exposures and myopic shift used for Figure 5.
|
Reference number |
Author (Year) |
Type |
N of dives |
Pressure (atm) |
Fraction of Oxygen |
Time (hours) |
Exposure |
N of |
Myopic shift |
Comments |
|
15 |
Fledelius (2002) |
HBOT |
30 |
2.50 |
0.95 |
1.58 |
112.81 |
17 |
0.62 |
|
|
10 |
Gesell (2007) |
HBOT |
48 |
2.50 |
1 |
1.50 |
180.00 |
1 |
3.375 |
After 11 months (no reversal) |
|
11 |
Hagan (2019) |
HBOT |
46 |
2.00 |
1 |
1.50 |
138.00 |
1 |
1.25 |
|
|
17 |
Anderson (1978) |
HBOT |
40 |
2.00 |
0.95 |
2.00 |
152.00 |
10 |
1.61 |
|
|
18 |
Palmquist (1984) |
HBOT |
1 |
2.25 |
1 |
426.33 |
959.24 |
25 |
3.00 |
The average number of treatment hours was calculated from the article and multiplied by 2.25 atm as the pressure was give 2−2.5atm |
|
20 |
Riedl (2019) |
HBOT |
40 |
2.40 |
1 |
1.50 |
144.00 |
29 |
0.705 |
|
|
21 |
Evanger (2015) |
HBOT |
20 |
2.40 |
1 |
1.50 |
72.71* |
20 |
0.63 |
|
|
22 |
Evanger (2018) |
HBOT |
30 |
2.40 |
1 |
1.50 |
108.71* |
23 |
0.95 |
|
|
23 |
Evanger (2004) |
HBOT |
21 |
2.40 |
1 |
1.50 |
76.31* |
20 |
0.54 |
Mask group |
|
23 |
Evanger (2004) |
HBOT |
21 |
2.40 |
1 |
1.50 |
76.31* |
12 |
1.08 |
Hood group |
|
24 |
Evanger (2014) |
HBOT |
20 |
2.40 |
1 |
1.50 |
72.71* |
15 |
0.62 |
|
|
12 |
Evanger (2006) |
HBOT |
21 |
2.40 |
1 |
1.50 |
75.60 |
1 |
1.31 |
|
|
26 |
Bennett (2019) |
HBOT |
20 |
2.40 |
1 |
1.50 |
72.00 |
55 |
0.92 |
Hood group, 20 treatments |
|
26 |
Bennett (2019) |
HBOT |
20 |
2.40 |
1 |
1.50 |
72.00 |
49 |
0.52 |
Mask group, 20 treatments |
|
26 |
Bennett (2019) |
HBOT |
30 |
2.40 |
1 |
1.50 |
108.00 |
42 |
1.25 |
Hood group, 30 treatments |
|
26 |
Bennett (2019) |
HBOT |
30 |
2.40 |
1 |
1.50 |
108.00 |
37 |
0.63 |
Mask group, 30 treatments |
|
14 |
Butler (1999) |
SCUBA |
21 |
1.30 |
1 |
4.04 |
110.29 |
1 |
1.5 |
|
|
25 |
Fock (2013) |
SCUBA |
26 |
1.40 |
1 |
1.87 |
67.95 |
14 |
0.4 |
The exact number of dives is unclear but it was assumed to be 13 days of 2 dives per day. The partial pressure of oxygen was simplified as 1.4 atm throughout the dive. |
Sokolowski et al., Supplementary File 5. Case report oxygen exposure
|
Dive time (min) |
Average depth (m) |
Nitrox used (O2%) |
Exposure (hours at 101 kPa) |
|
74 |
15 |
32 |
0.99 |
|
66 |
15 |
32 |
0.88 |
|
71 |
13 |
21 |
0.57 |
|
61 |
8 |
21 |
0.38 |
|
71 |
16 |
21 |
0.65 |
|
72 |
17 |
32 |
1.04 |
|
71 |
17 |
32 |
1.02 |
|
49 |
22 |
32 |
0.84 |
|
68 |
18 |
32 |
1.02 |
|
62 |
17 |
32 |
0.89 |
|
69 |
7 |
32 |
0.63 |
|
64 |
19 |
32 |
0.99 |
|
71 |
17 |
32 |
1.02 |
|
70 |
17 |
32 |
1.01 |
|
68 |
17 |
32 |
0.98 |
|
73 |
17 |
32 |
1.05 |
|
70 |
17 |
32 |
1.01 |
|
68 |
17 |
32 |
0.98 |
|
71 |
17 |
32 |
1.02 |
|
71 |
17 |
32 |
1.02 |
|
71 |
18 |
32 |
1.06 |
|
71 |
17 |
32 |
1.02 |
|
67 |
16 |
32 |
0.93 |
|
62 |
12 |
32 |
0.73 |
|
73 |
17 |
32 |
1.05 |
|
70 |
16 |
32 |
0.97 |
|
68 |
18 |
32 |
1.02 |
|
65 |
10 |
32 |
0.69 |
|
71 |
17 |
32 |
1.02 |
|
72 |
17 |
32 |
1.04 |
|
70 |
15 |
32 |
0.93 |
|
62 |
12 |
32 |
0.73 |
|
72 |
18 |
32 |
1.08 |
|
70 |
16 |
32 |
0.97 |
|
66 |
17 |
32 |
0.95 |
|
93 |
9 |
32 |
0.94 |
|
62 |
19 |
32 |
0.96 |
|
72 |
17 |
32 |
1.04 |
|
62 |
13 |
32 |
0.76 |
|
71 |
16 |
32 |
0.98 |
|
70 |
17 |
32 |
1.01 |
|
65 |
18 |
32 |
0.97 |
|
60 |
9 |
32 |
0.61 |
|
70 |
15 |
32 |
0.93 |
|
67 |
18 |
32 |
1.00 |
|
60 |
18 |
32 |
0.90 |
|
67 |
15 |
32 |
0.89 |
|
66 |
13 |
32 |
0.81 |
|
73 |
17 |
32 |
1.05 |
|
62 |
10 |
32 |
0.66 |
|
63 |
19 |
32 |
0.97 |
|
71 |
17 |
32 |
1.02 |
|
60 |
19 |
32 |
0.93 |
|
67 |
11 |
32 |
0.75 |
|
64 |
18 |
32 |
0.96 |
|
70 |
17 |
32 |
1.01 |
|
68 |
16 |
32 |
0.94 |
|
66 |
14 |
32 |
0.84 |
|
67 |
18 |
32 |
1.00 |
|
69 |
16 |
32 |
0.96 |
|
64 |
12 |
32 |
0.75 |
|
69 |
16 |
32 |
0.96 |
|
70 |
18 |
32 |
1.05 |
|
67 |
14 |
32 |
0.86 |
|
67 |
12 |
32 |
0.79 |
|
72 |
15 |
32 |
0.96 |
|
68 |
18 |
32 |
1.02 |
|
78 |
11 |
32 |
0.87 |
|
70 |
8 |
32 |
0.67 |
|
66 |
18 |
32 |
0.99 |
|
67 |
17 |
32 |
0.96 |
|
67 |
17 |
32 |
0.96 |
|
51 |
13 |
32 |
0.63 |
|
68 |
18 |
32 |
1.02 |
|
71 |
18 |
32 |
1.06 |
|
Total exposure (hours at 101 kPa) |
68.60 |
||
- Super User
- Category: Information
- 134
- Super User
- Category: Information
- 905
CHEERS Checklist
Section |
Item No |
Recommendation |
Reported on page No |
| Title and abstract | |||
| Title | 1 | Identify the study as an economic evaluation or use more specific terms such as “cost-effectiveness analysis”, and describe the interventions compared. | 1,2 |
| 2 | Provide a structured summary of objectives, perspective, setting, methods (including study design and inputs), results (including base case and uncertainty analyses), and conclusions. | 2 | |
| Introduction | |||
| Background and objectives | 3 | Provide an explicit statement of the broader context for the study. Present the study question and its relevance for health policy or practice decisions. | 4&5 |
| Methods | |||
| Target population and subgroups | 4 | Describe characteristics of the base case population and subgroups analysed, including why they were chosen. | 6 |
| Setting and location | 5 | State relevant aspects of the system(s) in which the decision(s) need(s) to be made. | 6 |
| Study perspective | 6 | Describe the perspective of the study and relate this to the costs being evaluated. | 7 |
| Comparators | 7 | Describe the interventions or strategies being compared and state why they were chosen. | 7 |
| Time horizon | 8 | State the time horizon(s) over which costs and consequences are being evaluated and say why appropriate. | 7 |
| Discount rate | 9 | Report the choice of discount rate(s) used for costs and outcomes and say why appropriate. | 7 |
| Choice of health outcomes | 10 | Describe what outcomes were used as the measure(s) of benefit in the evaluation and their relevance for the type of analysis performed. | 7-8 |
| Measurement of effectiveness | 11a | Single study-based estimates: Describe fully the design features of the single effectiveness study and why the single study was a sufficient source of clinical effectiveness data. | 7-10 |
| 11b | Synthesis-based estimates: Describe fully the methods used for identification of included studies and synthesis of clinical effectiveness data. | N.A | |
| Measurement and valuation of preference based outcomes | 12 | If applicable, describe the population and methods used to elicit preferences for outcomes. | 7-9 |
| Estimating resources and costs | 13a | Single study-based economic evaluation: Describe approaches used to estimate resource use associated with the alternative interventions. Describe primary or secondary research methods for valuing each resource item in terms of its unit cost. Describe any adjustments made to approximate to opportunity costs. | 8 |
| 13b | Model-based economic evaluation: Describe approaches and data sources used to estimate resource use associated with model health states. Describe primary or secondary research methods for valuing each resource item in terms of its unit cost. Describe any adjustments made to approximate to opportunity costs. | N.A. | |
| Currency, price date and conversion | 14 | Report the dates of the estimated resource quantities and unit costs. Describe methods for adjusting estimated unit costs to the year of reported costs if necessary. Describe methods for converting costs into a common currency base and the exchange rate. | 8 |
| Choice of model | 15 | Describe and give reasons for the specific type of decision analytical model used. Providing a figure to show model structure is strongly recommended. | 7-10 |
| Assumptions | 16 | Describe all structural or other assumptions underpinning the decision-analytical model. | 7-10 |
| Analytical methods | 17 | Describe all analytical methods supporting the evaluation. This could include methods for dealing with skewed, missing, or censored data; extrapolation methods; methods for pooling data; approaches to validate or make adjustments (such as half cycle corrections) to a model; and methods for handling population heterogeneity and uncertainty. | 9-10 |
| Results | |||
| Study parameters | 18 | Report the values, ranges, references, and, if used, probability distributions for all parameters. Report reasons or sources for distributions used to represent uncertainty where appropriate. Providing a table to show the input values is strongly recommended. | 11-12 |
| Incremental costs and outcomes | 19 | For each intervention, report mean values for the main categories of estimated costs and outcomes of interest, as well as mean differences between the comparator groups. If applicable, report incremental cost-effectiveness ratios. | 11-12 |
| Characterising uncertainty | 20a | Single study-based economic evaluation: Describe the effects of sampling uncertainty for the estimated incremental cost and incremental effectiveness parameters, together with the impact of methodological assumptions (such as discount rate, study perspective). | 11-12 |
| 20b | Model-based economic evaluation: Describe the effects on the results of uncertainty for all input parameters, and uncertainty related to the structure of the model and assumptions. | N.A. | |
| Characterising heterogeneity | 21 | If applicable, report differences in costs, outcomes, or cost-effectiveness that can be explained by variations between subgroups of patients with different baseline characteristics or other observed variability in effects that are not reducible by more information. | N.A |
| Discussion | |||
| Study findings, limitations, generalisability, and current knowledge | 22 | Summarise key study findings and describe how they support the conclusions reached. Discuss limitations and the generalisability of the findings and how the findings fit with current knowledge. | 13-15 |
| Other | |||
| Source of funding | 23 | Describe how the study was funded and the role of the funder in the identification, design, conduct, and reporting of the analysis. Describe other non-monetary sources of support. | 1 |
| Conflict of interest | 24 | Describe any potential for conflict of interest of study contributors in accordance with journal policy. In the absence of a journal policy, we recommend authors comply with International Committee of Medical Journal Editors recommendations. | 1 |