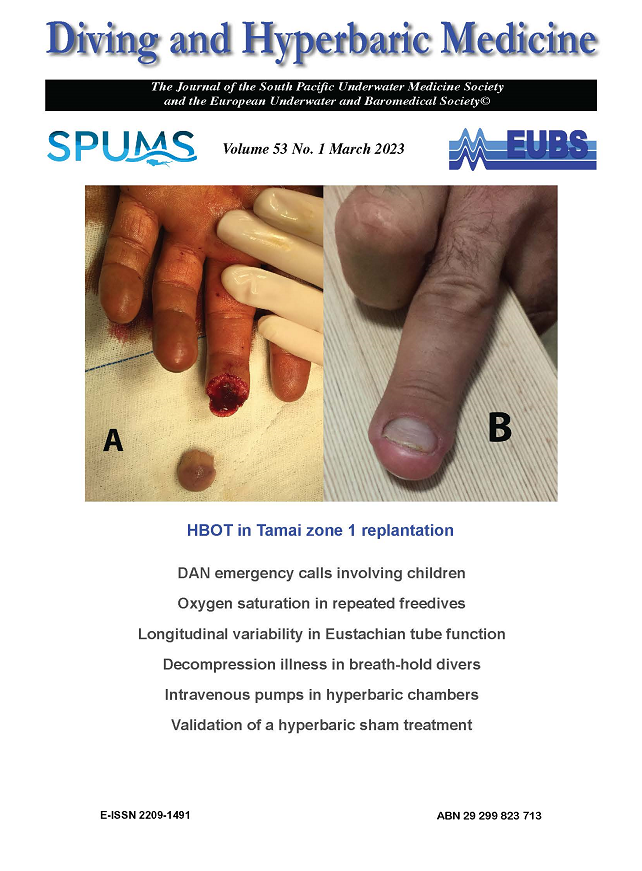
52 - 55
- Super User
- Category: Information
- 6380
Draft
Equipoise: an important ethical consideration when contemplating participation in a randomised controlled trial of hyperbaric oxygen treatment in necrotising soft tissue infections
Bridget Devaney1
1 Department of Intensive Care and Hyperbaric Medicine, Alfred Health, Melbourne, Australia
Corresponding author: Dr Bridget Devaney, Head of Hyperbaric Medicine, Alfred Health, 55 Commercial Road, Melbourne, VIC 3004, Australia
ORCID ID: 0000-0001-6521-418X
Keywords
Clinical Trials; Ethics; Hyperbaric research
Abstract
(Devaney B. Equipoise: an important ethical consideration when contemplating participation in a randomised controlled trial of hyperbaric oxygen treatment in necrotising soft tissue infections. Diving and Hyperbaric Medicine. 2024 31 March;54(1):NN−nn. doi: 10.28920/dhm54.1.NN-nn. PMID:????)
A proposal for a large, multi-centre, randomised controlled trial investigating the role of hyperbaric oxygen treatment (HBOT) in necrotising soft tissue infections (NSTI) has led to much discussion locally and internationally about whether participation is ethical for a centre where stakeholders already consider HBOT standard practice. This article systematically addresses the concept of clinical equipoise specific to the role of HBOT in NSTI, and presents a series of considerations to be taken into account by key stakeholders at potential participating sites.
Introduction
Highly regarded and widely published Danish colleagues are in the advanced phases of planning a multinational randomised controlled trial (RCT) investigating the use of hyperbaric oxygen treatment (HBOT) for necrotising soft tissue infections (NSTI). The trial involves patients with NSTI being randomised to receive either standard care (surgical debridement, antibiotics, intensive care support) or standard care plus HBOT.
Some Australian hospitals have used HBOT as part of the treatment for NSTI for many years, based on early work in clostridial infections, clinical experience and several retrospective and prospective studies that indicate HBOT may provide a mortality benefit.1–3
There has not previously been an RCT addressing this. Challenges in planning an RCT are imposed by the rarity of NSTI (requiring a multicentre, international study to achieve adequate power), the practical issues and considerations for the management of severely ill patients, as well as uncertainty amongst stakeholders regarding the presence or absence of equipoise for the role of HBOT in the treatment of NSTI.
Equipoise is a state of genuine uncertainty regarding the role of a treatment modality or the superiority of one treatment over another. It is a fundamental requirement of ethical clinical research, seeking to first do no harm. Equipoise may exist in an individual clinician who is indifferent to the treatment modalities (‘individual equipoise’) or amongst the expert medical community where ‘honest professional disagreement’ exists regarding the role of a treatment, or regarding which treatment modality is best (‘clinical equipoise’).4,5 A reliance on individual equipoise of all clinician investigators in a trial, presents potentially insurmountable obstacles to the commencement or completion of a controlled trial, and the impact of such a scenario on an RCT for HBOT in NSTI will be discussed below. In contrast, clinical equipoise considers the entire range of expert medical opinion as a priori equally valuable; essentially constituting a ‘fair bet’ procedure – and as such RCTs in areas of clinical equipoise are considered to not present a risk of harm to trial participants.6
In this article I will endeavour to systematically address the concept of clinical equipoise specific to the role of HBOT in NSTI.
Commentary
The critical question is ‘is it ethical for centres which already utilise HBOT as an adjunct to standard treatment for NSTI, to be involved in a study where fifty percent of patients will be randomised to not receive HBOT? The answer involves another question (a few, actually):
IS HBOT STANDARD TREATMENT FOR NSTI?
My health service treats more cases of NSTI with HBOT per year than all other Australian and New Zealand centres combined, so this question requires particular consideration.7 To adequately answer the question, we need to review whether the provision of HBOT for NSTI is considered standard practice at the individual clinician level, Health Service level, State level, national level, and also at an international level.
IS TREATMENT OF NSTI WITH HBOT CONSISTENTLY OFFERED BY ALL CLINICIANS AT YOUR HEALTH SERVICE?
Or does the provision of HBOT depend on specific clinicians being present, rostered on, and aware of an NSTI case in your centre (e.g., an anaesthetist who is also a hyperbaric physician being made aware of the case in theatre, hyperbaric doctors ‘finding’ cases, or a ‘believer’ specialty doctor making a referral to the Hyperbaric Service)?
In centres where there is variability between clinicians, patients with NSTI are essentially already receiving ‘random’ care (e.g., receiving HBOT or not, based on factors independent of any evidence). In this case, it is roster allocations or plain chance that determine the treatment pathway the patient is allocated to, without the advantages of an RCT to advance the level of evidence for (or against) this practice. Participating in an RCT simply changes the mode of allocation of treatment that is already occurring in many centres (amongst numerous other advantages).
IS THERE A CONSENSUS AMONGST CLINICIANS AT YOUR HEALTH SERVICE ABOUT THE ROLE OF HBOT FOR NSTI?
In Melbourne, we treat more cases of NSTI with HBOT than any other centre in Australia or New Zealand.7 Despite this, there is still a lack of consensus about the role of HBOT for NSTI.
Indeed, we evaluated this specific question and published our findings in ANZ Journal of Surgery in 2021.8 We surveyed experts at our centre on their beliefs about the role of HBOT in the treatment of NSTI. Whilst some clinicians felt strongly (n = 4, 6% strongly disagreed that HBOT has a role in the treatment of NSTI and n = 8, 12% strongly agreed), the most common response (n = 31, 45%) was not being sure if HBOT has a role in the treatment of NSTI. We concluded that there is clinical equipoise at our centre regarding the role of HBOT in the treatment of NSTI, that an RCT should be considered ethical, and that further work towards increasing the level of evidence is highly necessary.
ARE PATIENTS WITH NSTI ROUTINELY OFFERED HBOT IN YOUR STATE?
In Victoria, Austraila, they are not. Results from a (currently unpublished) project in which data from the Victorian admitted episodes dataset (VAED) and the Australia and New Zealand Intensive Care Society (ANZICS) adult patient database (APD) were linked by the Centre of Victorian Data Linkage (CVDL), indicate that less than one third of NSTI patients admitted to intensive care units in Victoria receive HBOT. That means that over two-thirds of Victorians who develop NSTI are not currently being referred for or receiving HBOT. Of interest, no statistically significant difference was found in APACHE III score or predicted risk of death in the groups who went on to receive, or not receive, HBOT.
WHAT ABOUT ON A REGIONAL LEVEL? IS HBOT FOR NSTI CONSIDERED STANDARD ACROSS AUSTRALIA & NEW ZEALAND?
It is not. Table 1 contains the number of cases of NSTI who received HBOT as reported by each Hyperbaric facility around Australasia in the 2022-2023 financial year; if the Alfred’s case numbers reflect less than one third of the Victorian NSTI case load, these statistics indicate that only a very small fraction of patients from around Australasia are currently receiving HBOT for NSTI. Assuming that disease incidence is similar across Australia and New Zealand, these data indicate a greater than ten-fold variation in the use of HBOT between regions.9,10
Table 1
Cases of necrotising soft tissue infections treated with hyperbaric oxygen in Australia and New Zealand (NZ); ACT – Australian Capital Territory; NSW – New South Wales;
NT – Northern Territory; pop – population; QLD – Queensland; SA – South Australia; TAS – Tasmania; VIC – Victoria; WA – Western Australia
|
Institution |
The Alfred Hospital (VIC) |
Fiona Stanley Hospital |
Royal Hobart Hospital |
Royal Adelaide Hospital |
Prince of Wales Hospital |
Royal Brisbane & Women’s Hospital |
Wesley Hospital |
Townsville |
Royal Darwin Hospital |
North Shore Hospital Auckland |
Christ-church Hospital |
|
Cases treated4 |
27 |
5 |
3 |
1 |
6 |
1 |
0 |
1 |
0 |
2 |
0 |
|
Population6,7 |
6,766,600 |
2,855,600 |
572,700 |
1,844,600 |
8,758,600 |
5,418,500 |
251,700 |
5,223,100 |
|||
|
Cases treated per 106 pop. |
3.99 |
1.75 |
5.24 |
0.54 |
0.69 |
0.37 |
0.00 |
0.38 |
|||
WHAT ABOUT ON AN INTERNATIONAL LEVEL? IS HBOT STANDARD PRACTICE FOR NSTI INTERNATIONALLY?
It is not. The use of HBOT for NSTI varies markedly between countries.
In July 2018 the NHS England published their Clinical Commissioning Policy: Hyperbaric Oxygen Therapy for necrotising soft tissue infections (all ages).11 They concluded that there is not enough evidence to make the treatment available at this time, and funding was removed for the use of HBOT for NSTI from 1 April 2019. Likewise in the USA, only ~ 1% of NSTI cases are treated with HBOT.12
In contrast, more than one third of patients with NSTI in Denmark receive HBOT.3
DO INTERNATIONAL SCIENTIFIC SOCIETIES UNIVERSALLY RECOMMEND HBOT FOR NSTI?
The recommendations from international societies vary; some do not recommend HBOT (e.g., The American Infectious Disease Society),13 some do recommend HBOT (e.g., The European and American Societies for diving and hyperbaric medicine),14,15 and some suggest consideration of HBOT if available and not interfering with standard treatment (e.g., World Society of Emergency Surgery and the Surgical Infection Society Europe).16
WHAT DOES COCHRANE SAY?
The authors of a Cochrane review published in 2015 concluded: “This systematic review failed to locate relevant clinical evidence to support or refute the effectiveness of HBOT in the management of necrotizing fasciitis. Good quality clinical trials are needed to define the role, if any, of HBOT in the treatment of individuals with necrotizing fasciitis”.17
SO DOES CLINICAL EQUIPOISE EXIST?
Irrefutably, at every level.
Nevertheless, one could argue that with all this uncertainty, maybe it will be simpler to just stay sitting on the fence? Definitely. This trial won’t be quick, or easy. However, without a unified effort, the likelihood of completion of this RCT falls. The status quo will remain; ongoing uncertainty amongst experts, ongoing inequity for patients, and ongoing inconsistency in the delivery of care for people with NSTI at hospital, state, national and international levels.
It is critical that such an RCT is planned by experts. If a poorly planned or inadequately powered trial were to be conducted, the outcome would likely be negative and may result in reduced use of HBOT for NSTI at centres which currently utilise HBOT, regardless of the actual impact HBOT has on NSTI. Clinical opinion may also shift away from a state of equipoise, which would reduce the possibility of a future, well conducted trial.
ARE THERE ANY OTHER ETHICAL CONSIDERATIONS WE SHOULD BE THINKING ABOUT?
I think it’s important to think about the ethics of not participating in a large, well-designed, multi-national, randomised controlled trial.
Our centre could take the position that HBOT is standard practice that would be unethical to withhold from 14 of the 28 Victorian patients with NSTI we treat on average per year.
However approximately 60 other Victorians are admitted to intensive care units with NSTI each year and are not referred for HBOT, no doubt in part because the current level of evidence isn’t considered adequately robust. There are hundreds of people around Australia who develop NSTI each year who do not receive HBOT, and there will be countless other people, around the world, who will develop NSTI into the future. When deciding whether or not to participate in an RCT, we must consider the large number of people into the future who this choice will impact.
Conclusion
A carefully designed, multi-centre, international randomised controlled trial investigating whether HBOT has a mortality benefit in patients with NSTI, has the potential for profound and lasting impact regardless of the outcome. A negative study may result in reduced workload of hyperbaric units around the world, millions of healthcare dollars saved and the substantial logistics involved with transferring patients with NSTI to hyperbaric services reduced. A positive study may impact the lives of thousands of NSTI sufferers into the future by resulting in increased use of HBOT and increased survival for these patients.
Without clearer answers, health services are unlikely to invest healthcare dollars into improving capacity for hyperbaric treatment of intensive care patients (which may already contribute to the low treatment numbers currently reported in many hyperbaric centres), and many NSTI patients will not be offered HBOT as a result.
If we do nothing, and maintain the status quo, only a small fraction of NSTI cases will receive HBOT at a state, national and international level. If there is a survival benefit from HBOT – which observational data suggest may be the case - remaining at status quo will do more harm than good. Perhaps the real question should be: is it ethical not to participate?
What’s your position?
References
- Devaney B, Frawley G, Frawley L, Pilcher DV. Necrotising soft tissue infections: the effect of hyperbaric oxygen on mortality. Anaesth Intensive Care. 2015;43:685–92. doi: 10.1177/0310057X1504300604. PMID: 26603791.
- Hedetoft M, Bennett MH, Hyldegaard O. Adjunctive hyperbaric oxygen treatment for necrotising soft-tissue infections: A systematic review and meta-analysis. Diving Hyperb Med. 2021;51:34–43. doi: 10.28920/dhm51.1.34-43. PMID: 33761539. PMCID: PMC8081587.
- Hedetoft M, Madsen MB, Madsen LB, Hyldegaard O. Incidence, comorbidity and mortality in patients with necrotising soft-tissue infections 2005–2018: a Danish nationwide register-based cohort study. BMJ Open. 2020;10(10)e041302. doi: 10.1136/bmjopen-2020-041302. PMID: 33067303. PMCID: PMC7569942.
- Nardini C. The ethics of clinical trials. Ecancermedicalscience. 2014;8:387. doi: 10.3332/ecancer.2014.387. PMID: 24482672. PMCID: PMC3894239.
- Freedman B. Equipoise and the ethics of clinical research. N Engl J Med. 1987;317(3):141–5. doi: 10.1056/NEJM198707163170304. PMID: 3600702.
- Edwards SJ, Lilford RJ, Braunholtz DA, Jackson JC, Hewison J, Thornton J. Ethical issues in the design and conduct of randomised controlled trials. Health Technol Assess. 1998;2(15):i–vi, 1–132. PMID: 10194615.
- Hyperbaric Technicians and Nurses Association. Australian and New Zealand Hyperbaric Treatment Data, 1 July 2022 – 30 June 2023. HTNA 2023.
- Devaney B, Pilcher D, Mitra B, Watterson J. Does equipoise exist amongst experts regarding the role of hyperbaric oxygen treatment for necrotising soft tissue infection? ANZ J Surg. 2021;91:485–7. doi: 10.1111/ans.16337. PMID: 33847053.
- Australian Bureau of Statistics. National, state and territory population. Reference period: March 2023. [cited 2023 Oct 3]. Available from: https://www.abs.gov.au.
- Statistics New Zealand. Tatauranga Aotearoa. National population estimates: At 30 June 2023. [cited 2023 Oct 3]. Available from: https://www.stats.govt.nz.
- NHS England. Clinical Commissioning Policy: Hyperbaric Oxygen Therapy for necrotising soft tissue infections (all ages). July 2018. [cited 2023 Oct 3]. Available from: https://www.england.nhs.uk/wp-content/uploads/2018/07/hbot-for-necrotising-soft-tissue-infections-v2.pdf.
- Soh CR, Pietrobon R, Freiberger JJ, Chew ST, Rajgor D, Gandhi M, et al. Hyperbaric oxygen therapy in necrotising soft tissue infections: a study of patients in the United States Nationwide Inpatient Sample. Intensive Care Med. 2012;38:1143–51. doi: 10.1007/s00134-012-2558-4. PMID: 22527074.
- Stevens DL, Bisno AL, Chambers HF, Dellinger EP, Goldstein EJ, Gorbach SL, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the infectious diseases society of America. Clin Infect Dis. 2014;59:147–59. doi: 10.1093/cid/ciu296. PMID: 24947530.
- Mathieu D, Marroni A, Kot J. Tenth European Consensus Conference on Hyperbaric Medicine: recommendations for accepted and non-accepted clinical indications and practice of hyperbaric oxygen treatment. Diving Hyperb Med. 2017;47:24–32. doi: 10.28920/dhm47.1.24-32. PMID: 28357821. PMCID: PMC6147240.
- Moon RE, editor. Undersea and Hyperbaric Medical Society. Hyperbaric oxygen therapy indications. 14th ed. North Palm Beach (FL): Best Publishing Company; 2019.
- Sartelli M, Coccolini F, Kluger Y, Agastra E, Abu-Zidan FM, Abbas AES, et al. WSES/GAIS/WSIS/SIS-E/AAST global clinical pathways for patients with skin and soft tissue infections. World J Emerg Surg. 2022;17(1):1–23. doi: 10.1186/s13017-022-00406-2. PMID: 35033131. PMCID: PMC8761341.
- Levett D, Bennett MH, Millar I. Adjunctive hyperbaric oxygen for necrotizing fasciitis. Cochrane Database Syst Rev. 2015;1(1):CD007937. doi: 10.1002/14651858.CD007937.pub2. PMID: 25879088. PMCID: PMC6516968.
Conflicts of interest and funding: nil
Submitted: 17 November 2023
Accepted after revision: 19 January 2024
Copyright: This article is the copyright of the author who grants Diving and Hyperbaric Medicine a non-exclusive licence to publish the article in electronic and other forms
- Super User
- Category: Information
- 1637
Draft
Reported outcome measures in necrotising soft tissue infections: a systematic review
Jonathan Wackett1, Bridget Devaney2,3, Raymond Chau4, Joshua Ho4, Nicholas King4, Jasleen Grewal1, Joshua Armstrong5, Biswadev Mitra2,6
1 Department of Medicine, Alfred Health, Melbourne, Australia
2 Emergency and Trauma Centre, Alfred Health, Melbourne, Australia
3 Department of Intensive Care and Hyperbaric Medicine, Alfred Health, Melbourne, Australia
4 Monash Health, Melbourne, Australia
5 Barwon Health, Geelong, Australia
6 Department of Epidemiology and Preventative Medicine, Monash University, Melbourne, Australia
Corresponding author: Dr Bridget Devaney, Head of Hyperbaric Medicine, Alfred Health, 55 Commercial Road, Melbourne, VIC 3004, Australia
ORCID ID: 0000-0001-6521-418X
Keywords
Gas gangrene; Hyperbaric oxygen treatment; Intensive care medicine; Systematic review
Abstract
(Wackett J, Devaney B, Chau R, Ho J, King N, Grewal J, Armstrong J, Mitra B. Reported outcome measures in necrotising soft tissue infections: a systematic review. Diving and Hyperbaric Medicine. 2024 31 March;54(1):NN−nn. doi: 10.28920/dhm54.1.NN-nn. PMID:????)
Introduction: There are inconsistencies in outcome reporting for patients with necrotising soft tissue infections (NSTI). The aim of this study was to evaluate reported outcome measures in NSTI literature that could inform a core outcome set (COS) such as could be used in a study of hyperbaric oxygen in this indication.
Methods: A systematic review of all NSTI literature identified from Cochrane, Ovid MEDLINE and Scopus databases as well as grey literature sources OpenGrey and the New York Academy of Medicine databases which met inclusion criteria and were published between 2010 and 2020 was performed. Studies were included if they reported on > 5 cases and presented clinical endpoints, patient related outcomes, or resource utilisation in NSTI patients. Studies did not have to include intervention. Two independent researchers then extracted reported outcome measures. Similar outcomes were grouped and classified into domains to produce a structured inventory. An attempt was made to identify trends in outcome measures over time and by study design.
Results: Three hundred and seventy-five studies were identified and included a total of 311 outcome measures. Forty eight percent (150/311) of outcome measures were reported by two or more studies. The four most frequently reported outcome measures were mortality without time specified, length of hospital stay, amputation performed, and number of debridements, reported in 298 (79.5%), 260 (69.3%), 156 (41.6%) and 151 (40.3%) studies respectively. Mortality outcomes were reported in 23 different ways. Randomised controlled trials (RCTs) were more likely to report 28-day mortality or 90-day mortality. The second most frequent amputation related outcome was level of amputation, reported in 7.5% (28/375) of studies. The most commonly reported patient-centred outcome was the SF-36 which was reported in 1.6% (6/375) of all studies and in 2/10 RCTs.
Conclusions: There was wide variance in outcome measures in NSTI studies, further highlighting the need for a COS.
Introduction
Necrotising soft tissue infections (NSTI) are a collection of rare but serious infections that can lead to widespread tissue destruction and threaten considerable morbidity and mortality. NSTI encompasses conditions such as necrotising fasciitis, Fournier’s gangrene, necrotising cellulitis and necrotising myonecrosis.1 A large Danish registry-based study demonstrated all-cause mortality rates of 19% at 30 days, 25% at 90-days, and 30% at one-year.2 Treatment modalities include early surgical debridement, broad spectrum antibiotics and often organ support in an intensive care unit, however there is ongoing discourse as to the effectiveness of adjuvant therapies such as hyperbaric oxygen treatment (HBOT) and intravenous immunoglobulin (IVIG) administration.1
Treatment with hyperbaric oxygen involves breathing 100% oxygen at greater than atmospheric pressures, substantially increasing serum partial pressures of oxygen. There are a number of proposed phyiological mechanisms by which repeated increased partial pressures of oxygen may improve outcomes in NSTI. Multiple retrospective observational studies and a recent meta-analysis demonstrate reduced in-hospital mortality in NSTI patients treated with HBOT, however Level 1 evidence is currently lacking and the use of HBOT varies between centres.3 Heterogeneity in outcome reporting limits the quality of data available for meta-analysis.
Thus, it follows that the selection of outcome measures for prospective trials is critical.4 A core outcome set (COS) is an agreed standardised set of outcomes that should be measured and reported, as a minimum, in all clinical trials in specific areas of health or health care.4 High quality prospective trials use outcomes that are predetermined, but in the absence of a COS, findings are variably reported, and reporting bias may be introduced.5 Currently, there is no consensus amongst clinicians, researchers and patients regarding the outcome measures that should be collected and reported in studies assessing potential interventions for NSTI.6 A Cochrane Review of interventions for NSTIs in adults demonstrated that only one third of included studies reported all the predetermined outcomes.7 Such inconsistencies preclude the synthesis of data in meta-analyses and reduce the quality of evidence available to form clinically relevant conclusions that ultimately benefit patient care.
The aim of this systematic review is to develop an inventory of outcome measures used in NSTI studies. We evaluated associations between methodological design and outcome reporting. It was expected that the findings would inform the development of a COS for NSTI, which will lead to enhanced ability to evaluate the efficacy of adjuvant therapies such as HBOT.
Methods
This systematic review was designed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline.8 The review protocol was developed a priori and registered with the International Prospective Register of Systematic Reviews (PROSPERO), registration number CRD42022330268. The inventory of reported outcome measures generated by this review will inform Delphi surveys and consensus meetings as part of a broader initiative to develop COS in NSTI.
After the initial search, all steps were undertaken in duplicate by independent reviewers.
SEARCH STRATEGY
A comprehensive literature search was undertaken using Cochrane, Ovid MEDLINE and Scopus databases as well as grey literature sources OpenGrey and the New York Academy of Medicine databases. Searches were performed for a 10-year period (January 2010 - August 2020) to record an extensive list of outcomes being reported for this relatively rare group of conditions, as well as to identify how research in NSTI may have changed over time. Medical subject headings and keywords such as “Necrotising soft tissue infections”, “Fournier gangrene”, and “Gas gangrene” were combined using the “OR” operator to ensure a breadth of results were returned. An example of the full search strategy as was used for Ovid MEDLINE is provided in Appendix 1
STUDY ELIGIBILITY
Studies were included if they related to NSTI and reported one or more patient outcomes provided they also met the following criteria:
Types of studies: All study designs were included except for case reports, case series of < 5 cases, case series that only express their outcomes individually or qualitatively (e.g., a case series of 8 cases described in detail but with no pooling or tabulation of patient outcomes). These study designs were excluded to avoid outcome measures that are less relevant or achievable for larger studies.
Types of participants: We included studies that reported outcomes of NSTI patients of all ages, geographic locations, and disease phenotypes (necrotising fasciitis, Fournier’s gangrene etc) that were at any stage in the course of their disease (inpatient or outpatient).
Types of interventions: Studies of any/all interventions for NSTI were included. Studies not assessing an intervention were also included, provided they reported on patient outcomes.
Types of outcomes: Studies were included if they reported any patient related outcome or clinical endpoint, including outcomes related to mortality, morbidity, recovery, quality of life, and adverse events. Outcomes reported in the body of text, tables and/or figures were included. Patient and observer reported outcomes were included. Studies that did not include any patient centred outcomes or resource utilisation outcomes were excluded (e.g. laboratory-based studies reporting specific biomarkers only).
STUDY SELECTION PROCESS
All reviewers involved in the study selection process underwent training to ensure they understood the context of the review, the inclusion/exclusion criteria and how to use the Covidence software prior to study screening.
The title and abstract of each study were screened independently and in duplicate by two reviewers (BD, JA, JW). The primary reason for exclusion at this stage was study design (e.g., case study or case series with < 5 cases). The full text of studies found to meet the inclusion criteria were then retrieved. Again, two reviewers (JA, JG, JH, JW, RC) reviewed each study independently and in duplicate. Disputes at either stage were reviewed and resolved by the senior reviewer (JW).
QUALITY ASSESSMENT
Examination and synthesis of data related to patients or treatment effects was not performed. To produce an exhaustive list of outcomes and to compare potential differences in reporting between different study designs, all relevant studies were included, regardless of methodology. Thus, no risk of bias or quality assessment of studies was performed, as we only sought to extract the relevant outcome measures that were reported in each study.
DATA EXTRACTION
Online software from Research Electronic Data Capture (REDCap) was used to extract and securely store data.9 Alongside the outcome measures reported by each study, we recorded each study’s author, year of publication, country it was primarily conducted in, study design and number of NSTI patients included. We noted whether studies declared sources of funding or potential sources of bias, although this data is not presented here.
Data were extracted from each study independently and in duplicate by two reviewers (JG, JH, JW, NK, RC). Both primary and secondary outcomes were recorded.
DATA ANALYSIS
Following extraction in duplicate, the two sets of data were exported into Microsoft Excel. Any discrepancies were flagged and reviewed by the senior reviewer (JW).
Outcomes that were similar but spelt or worded differently were reviewed by the senior reviewer to ensure the meaning was the same and subsequently merged, for example; “days in hospital” and “length of hospital stay (days)”. Many studies reported the same outcome measure but at different time points, such as; “mortality at 7 days”, “mortality at 3 months”, “in-hospital mortality”. In these cases, they were included as separate outcomes, as it is the intent of this study to identify the individual outcomes and time points that were considered important to researchers of NSTI. Ultimately, an individual list of outcomes that were reported by each study was generated. This list was used to create a comprehensive outcome inventory.
One group has developed a taxonomy for outcome measures to increase the efficiency of searching resources and databases by facilitating uniformity of outcome classification.10 This taxonomy has been adopted by the Core Outcome Measures in Effectiveness Trials (COMET) initiative as well as the Cochrane Linked Data Project.10 With this work in mind, the outcome measures identified in this systematic review were organised into eleven different outcome domains and then classified under five core areas based on their subject matter; mortality, physiological/clinical, resource use, life impact and adverse outcomes.
Results
The online search retrieved 4,256 titles and they were exported to the reference management tool EndNote X8 where 1,069 duplicates were removed.11 Remaining studies were input into the online systematic review software Covidence where a further 303 duplicates were identified and removed.12 After abstracts had been screened, 436 studies selected for full text review. Figure 1 outlines this process.
Figure 1
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram; outcome reporting of patients with NSTI
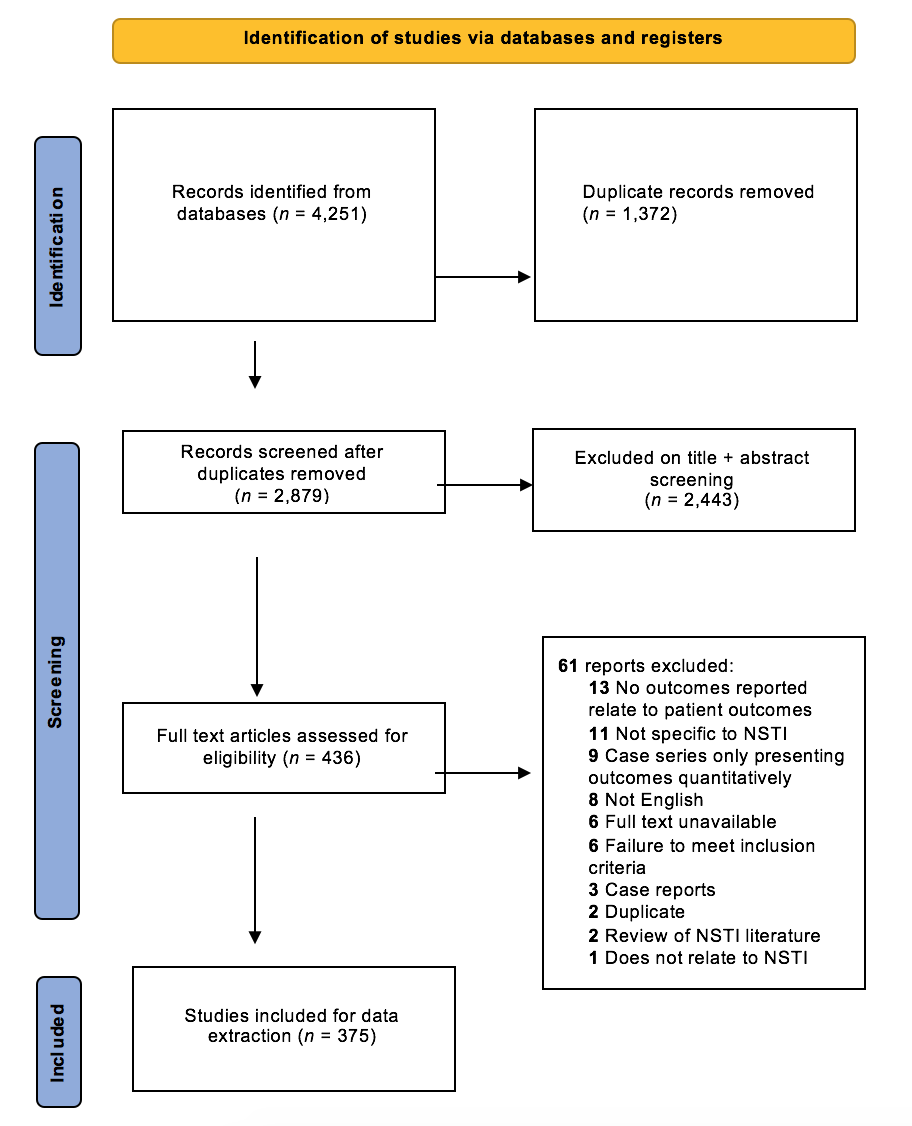
Three hundred and seventy-five studies were included; references are provided in Appendix 2. Of these, 86% (324/375) of studies were retrospective which included a total of 276,119 patients and 2.7% (10/375) were randomised controlled trials including 904 patients. In total, 7,062 patients were included in prospective studies.
A total of 311 distinct outcomes were reported 2,629 times by the included studies. Of these, 48% (150/311) of outcome measures were reported by two or more studies. Outcome measures were classified into 11 outcome domains and are presented under the five core areas consistent with the taxonomy developed elsewhere; mortality, physiological/clinical, life impact, resource use and adverse events.10 These are detailed below. A full inventory of the outcomes reported, their relative frequency and their stratified domains can be found in Appendix 3.
Tables 1 - 5 show the most reported outcomes in each domain. Each table outlines the total number of studies that reported an outcome and the number of patients in those studies. The total pool of studies is also further subdivided into study design, either prospective or randomised controlled trials (RCT) and by year of publication, either 2010–2015 or 2016–2020, to give an indication on how outcome reporting may be changing over time
CORE AREA: MORTALITY/SURVIVAL (TABLE 1)
Mortality without time specified was the most frequently reported mortality related outcome, appearing in 79.5% (298/375) of studies. 60% (6/10) of RCTs reported a mortality outcome, of which four specified a time point. 28-day mortality was the most commonly reported time point, appearing in 40% (4/10) of RCTs. 90-day mortality was more frequently reported in the second five-year period of extraction (2016-2020), being reported in 13/175 (7.4%) of included manuscripts. Survival time was less frequently reported among studies in the second 5-year period at 9.7% (17/175), compared to 8.5% (17/200) in the earlier period of this study. A further 16 mortality related outcomes can be found in Appendix 3.
Table 1
Mortality/survival outcomes; ICU – intensive care unit; RCTs – randomised controlled trials
|
Outcome |
% of total studies |
n patients |
Reported in % of prospective studies |
Reported in % of RCTs |
Reported in % of studies 2010–2015 |
Reported in % of studies 2016–2020 |
|
Mortality without time specified |
79.5% (298/375) |
267,990 |
45.1% (23/51) |
20% (2/10) |
85.5% (171/200) |
72.6% (127/175) |
|
In hospital mortality / 'survival to discharge' |
25.1% (94/375) |
11,3154 |
23.5% (12/51) |
20% (2/10) |
24.5% (49/200) |
25.7% (45/175) |
|
Survival time |
6.7% (25/375) |
48,607 |
11.7% |
0% (0/10) |
8.5% (17/200) |
4.6% (8/175) |
|
30-day mortality |
5.6% (21/375) |
10,184 |
21.6% (11/51) |
0% (0/10) |
4.5% (9/200) |
6.9% (12/175) |
|
28-day mortality |
5.3% (20/375) |
5,465 |
21.6% (11/51) |
40% (4/10) |
4.5% (9/200) |
6.3% (11/175) |
|
90-day mortality |
5.1% (19/375) |
1,900 |
29.4% (15/51) |
30% (3/10) |
3.0% (6/200) |
7.4% (13/175) |
|
ICU mortality |
3.5% (13/375) |
1,273 |
3.9% |
0% (0/10) |
3.0% (6/200) |
4.0% (7/175) |
CORE AREA: PHYSIOLOGICAL/CLINICAL (TABLE 2)
Amputation performed was an outcome reported in 41.6% (156/375) of studies and 50% (5/10) of RCTs. The next most reported amputation related outcome, level of amputation, was reported in 7.5% (28/375) of studies but was not recorded in any RCTs. There were 13 other amputation related outcomes were identified and can be found in Appendix 3, but none were reported by more than 3 studies.
Number of debridements required was the most reported debridement related outcome, being reported in 40.3% (151/375) of total studies, including 33.3% (17/51) of prospective studies and 40% (4/10) of RCTs. No other debridement related outcomes were reported in more than five studies. A further 10 debridement related outcomes can be found in Appendix 3.
Skin graft requirement was reported in 23.5% (88/375) of studies including 15.7% (8/51) of prospective studies. Surgical flap requirement (without regard to the specific type, e.g. rotational, free etc) was reported in 11.7% (44/375) of papers but only 2.0% (1/51) of prospective studies. There were 26 other closure/reconstruction outcomes Appendix 3.
Healing related outcomes. A total of 18 healing related outcomes were identified and can be found in Appendix 3. Only two, however, were reported by more than two studies. The most frequently recorded outcome was wound healing time (cicatrisation time) which could be found in 2.7% (10/375) of studies, including 316 patients.
Other Surgical outcomes. Number of procedures/surgeries required was recorded in 22.9% (86/375) of studies. Of those, 33.7% (29/86) also reported number of debridements required. There were 26 other surgical outcomes reported in Appendix 3.
Composite scores/endpoints. Numerous studies recorded sequential organ failure assessment (SOFA) scores at different stages of admission (e.g., score at day 1, day 2, day 7 etc). In an attempt to distinguish between patient characteristics and outcomes, the authors decided to include SOFA scores at time points longer than 14 days as outcomes. This juncture was chosen as the day-14 modified ‘mSOFA’ has been validated for NSTI patients as a part of the Necrotising Infection Clinical Composite Endpoint (NICCE).13 A total of 7 composite score outcomes are listed in Appendix 3, five of which were included in RCTs. The SOFA score (Day 14) was reported by 30% (3/10) of RCTs.
Table 2
Physiological/clinical outcomes; NICCE – necrotizing infection clinical composite endpoint; RCTs – randomised controlled trials; SOFA – sequential organ failure assessment
|
Outcome |
% of total studies |
n patients |
Reported in % of prospective studies |
Reported in % of RCTs |
Reported in % of studies 2010–15 |
Reported in % of studies 2016–20 |
|
Amputation outcomes |
||||||
|
Amputation performed |
41.6% (156/375) |
141,271 |
41.0% (25/51) |
50% (5/10) |
45% (90/200) |
37.7% (66/175) |
|
Level of amputation |
7.5% (28/375) |
8,133 |
7.8% (4/51) |
0% (0/10) |
9.5% (19/200) |
5.1% (9/175) |
|
Debridement outcomes |
||||||
|
Number of debridements required |
40.3% (151/375) |
110,122 |
33.3% (17/51) |
40% (4/10) |
37% (74/200) |
45.1% (79/175) |
|
Required debridement AND fasciotomy |
1.3% (5/375) |
362 |
0% (0/51) |
0% (0/10) |
1.5% (3/200) |
1.1% (2/175) |
|
Extent of debridement (cm2) |
1.1% (4/375) |
1,258 |
0% (0/51) |
0% (0/10) |
0.5% (1/200) |
1.7% (3/175) |
|
Closure/ reconstruction outcomes |
||||||
|
Skin graft requirement |
23.5% (88/375) |
8,129 |
15.7% (8/51) |
10% (1/10) |
25% (50/200) |
21.7% (38/175) |
|
Surgical flap requirement |
11.7% (44/375) |
2,628 |
2.0% (1/51) |
0% (0/10) |
11% (22/200) |
12.6% (22/175) |
|
Surgical reconstruction requirement |
10% (38/375) |
4,012 |
5.9% (3/51) |
10% (1/10) |
10% (20/200) |
10.3% (18/175) |
|
Primary wound closure |
5.9% (22/375) |
3,063 |
2.0% (1/51) |
0% (0/10) |
4% (8/200) |
8.0% (14/175) |
|
Split thickness skin graft requirement |
4.3% (16/375) |
808 |
2.0% (1/51) |
10% (1/10) |
5.5% (11/200) |
2.9% (5/175) |
|
Area healed by secondary intention |
2.7% (10/375) |
671 |
0% (0/51) |
0% (0/10) |
3.5% (7/200) |
1.7% (3/175) |
|
Healing outcomes |
||||||
|
Wound healing time (cicatrisation time) |
2.7% (10/375) |
316 |
5.9% (3/51) |
10% (1/10) |
2.5% (5/200) |
2.9% (5/175) |
|
Time to cure |
1.0% (3/375) |
315 |
0% (0/51) |
0% (0/10) |
1% (2/200) |
0.6% (1/175) |
|
Other surgical outcomes |
||||||
|
Number of operations required |
22.9% (86/375) |
7,1081 |
9.8% (5/51) |
0% (0/10) |
22.5% (45/200) |
23.4% (41/175) |
|
Colostomy |
20.0% (75/375) |
17,348 |
11.8% (6/51) |
10% (1/10) |
18.5% (37/200) |
21.7% (38/175) |
|
Orchidectomy |
12.3% (46/375) |
13,838 |
3.9% (2/51) |
10% (1/10) |
10.5% (21/200) |
14.3% (25/175) |
|
Cystostomy |
9.1% (34/375) |
2,288 |
7.8% (4/51) |
10% (1/10) |
9.5% (19/200) |
8.6% (15/175) |
|
Suprapubic tube placement |
5.6% (21/375) |
12,190 |
2.0% (1/51) |
0% (0/10) |
3.5% (7/200) |
8.0% (14/175) |
|
Penectomy |
4.0% (15/375) |
12,501 |
2.0% (1/51) |
0% (0/10) |
3.5% (7/200) |
4.6% (8/175) |
|
Faecal diversion |
3.2% (12/375) |
9,832 |
2.0% (1/51) |
0% (0/10) |
3.0% (6/200) |
3.4% (6/175) |
|
Composite outcomes |
||||||
|
SOFA score (Day 14) |
1.1% (4/375) |
821 |
5.9% (3/51) |
30% (3/10) |
1.5% (3/200) |
0.6% (1/175) |
|
NICCE endpoint |
0.8% (3/375) |
778 |
3.9% (2/51) |
20% (2/10) |
0.5% (1/200) |
1.1% (2/175) |
|
SOFA score (Day 28) |
0.5% (2/375) |
821 |
2.0% (1/51) |
10% (1/10) |
0.5% (1/200) |
0.6% (1/175) |
|
m-SOFA (Day 14) |
0.5% (2/375) |
488 |
2.0% (1/51) |
10% (1/10) |
0.5% (1/200) |
0.6% (1/175) |
CORE AREA: LIFE IMPACT (TABLE 3)
Patient perspective related outcomes. Outcomes relating to the patient’s perspective were recorded infrequently, with only four outcomes being reported by more than one study. The Medical Outcomes Short Form-36 questionnaire result (SF36) was the most reported patient perspective related outcome and was found in 1.6% (6/375) of all studies and was measured in 20% (2/10) of RCTs. Twenty more outcomes can be found in Appendix 3, all of which were only reported in one study each.
Table 3
Life impact outcomes; RCTs – randomised controlled trials
|
Outcome |
% of total studies |
n patients |
Reported in % of prospective studies |
Reported in % of RCTs |
Reported in % of studies 2010–15 |
Reported in % of studies 2016–20 |
|
Short Form-36 (SF36) |
1.6% (6/375) |
324 |
3.9% (2/51) |
20% (2/10) |
1% (2/200) |
2.3% (4/175) |
|
Pain score (visual analogue scale) |
0.5% (2/375) |
92 |
2.0% (1/51) |
20% (1/10) |
0% (0/200) |
1.1% (2/175) |
|
Derriford appearance scale |
0.5% (2/375) |
92 |
0% (0/51) |
0% (0/10) |
0.5% (1/200) |
0.6% (1/175) |
|
Disability |
0.5% (2/375) |
597 |
0% (0/51) |
0% (0/10) |
0% (0/200) |
1.1% (2/175) |
CORE AREA: RESOURCE USE (TABLE 4)
Length of hospital stay was reported in 69.3% (260/375) of studies, making it the second most commonly reported outcome overall after mortality without time specified. It was also reported in 47.1% (24/51) of prospective studies and 80% (8/10) of RCTs. Ventilation (days) was more frequently reported than ventilation (hours) appearing in 8.8% (33/375) of studies compared to 1.3% (5/375). There are 17 more resource use related outcomes listed in Appendix 3.
Discharge related outcomes. The most frequently reported discharge related outcome, discharge home, was reported in 4.8% (18/375) of studies, representing 40,466 patients. Discharge to skilled nursing facility was reported in 2.4% (9/375) of studies representing 113,368 patients. Nine further discharge related outcomes can be found in Appendix 3.
Table 4
Resource use outcomes; ICU – intensive care unit; RCTs – randomised controlled trials
|
Outcome |
% of total studies |
n patients |
Reported in % of prospective studies |
Reported in % of RCTs |
Reported in % of studies 2010–5 |
Reported in % of studies 2016–20 |
|
Resource outcomes |
||||||
|
Length of hospital stay |
69.3% (260/375) |
61,784 |
47.1% (24/51) |
80% (8/10) |
67.5% (135/200) |
71.4% (125/175) |
|
Length of ICU stay (days) |
27.5% (103/375) |
60,749 |
23.5% (12/51) |
40% (4/10) |
26% (52/200) |
29.1% (51/175) |
|
Ventilation (days) |
8.8% (33/375) |
4,127 |
13.7% (7/51) |
30% (3/10) |
9.5% (19/200) |
8.0% (14/175) |
|
Cost per patient |
2.9% (11/375) |
49,987 |
0% (0/51) |
0% (0/10) |
3.5% (7/200) |
2.3% (4/175) |
|
Ventilator-free days |
2.4% (9/375) |
11,730 |
7.8% (4/51) |
3% (3/10) |
2.5% (5/200) |
2.3% (4/175) |
|
Discharge outcomes |
||||||
|
Discharged home |
4.8% (18/375) |
40,466 |
5.9% (3/51) |
10% (1/10) |
4.5% (9/200) |
5.1% (9/175) |
|
Discharged to skilled nursing facility |
2.4% (9/375) |
113,368 |
2.0% (1/51) |
0% (0/10) |
1.5% (3/200) |
3.4% (6/175) |
|
Discharged to rehabilitation |
1.6% (6/375) |
1,576 |
2.0% (1/51) |
0% (0/10) |
2.5% (5/200) |
1.7% (3/175) |
|
Discharged to other hospital |
1.6% (6/375) |
10,237 |
0% (0/51) |
0% (0/10) |
1% (2/200) |
2.3% (4/175) |
|
Routine discharge |
1.6% (6/375) |
5,6151 |
2.0% (1/51) |
0% (0/10) |
2% (2/200) |
1.1% (2/175) |
CORE AREA: ADVERSE EVENTS (TABLE 5)
A total of 102 adverse event/complication outcomes are listed and further classified into subcategories in Appendix 3. Eighty-four of these were recorded in five or less studies.
Table 5
Adverse events outcomes; CVS – cardiovascular system; RCTs – randomised controlled trials
|
Outcome |
% of total studies |
n patients |
Reported in % of prospective studies |
Reported in % of RCTs |
Reported in % of studies 2010–15 |
Reported in % of studies 2016–20 |
|
Septic shock |
16.3% (61/375) |
60,019 |
15.7% (8/51) |
0% (0/10) |
13% (26/200) |
20% (35/175) |
|
Sepsis |
12.3% (46/375) |
13,215 |
7.8% (4/51) |
0% (0/10) |
13.5% (27/200) |
10.9% (19/175) |
|
Organ failure/ dysfunction |
11.2% (42/375) |
59,832 |
7.8% (4/51) |
20% (2/10) |
16% (32/200) |
5.7% (10/175) |
|
Acute kidney injury |
8.8% (33/375) |
10,037 |
7.8% (4/51) |
0% (0/10) |
8.5% (17/200) |
9.1% (16/175) |
|
Pneumonia |
6.1% (23/375) |
53,992 |
5.9% (3/51) |
10% (1/10) |
6.5% (13/200) |
5.7% (10/175) |
|
CVS complications (not otherwise spec) |
5.6% (21/375) |
116,920 |
3.9% (2/51) |
0% (0/10) |
5.5% (11/200) |
5.7% (10/175) |
|
Acute respiratory failure |
5.6% (21/375) |
63,160 |
2.0% (1/51) |
10% (1/10) |
6.5% (13/200) |
4.6% (8/175) |
Discussion
The major strength of this review is its comprehensive nature. A systematic and predetermined approach was utilised, and by using broad search terms, the studies identified are likely a thorough representation of the NSTI literature. All stages of the review were conducted in duplicate to reduce recording bias. To the best of the authors’ knowledge, this is the only study reporting systematically on outcome measures in contemporary NSTI literature. This review demonstrated variability in outcome reporting for NSTI. No single outcome was consistently found in every study and only four outcomes (mortality without time specified, length of hospital stay, amputation performed, number of debridements required) appeared in more than one third of studies. This heterogeneity of reporting limits evidence synthesis and the ability to compare data sets.14 Varied and inconsistent use of outcomes measures leaves meta-analyses unable to include data from all relevant studies or forces them to make assumptions about unclear reporting.7,15
Studies representing less than five patients were excluded from this review, as were those that made no attempt to summarise or pool their results. Therefore, it is probable that certain novel or unique NSTI outcomes were missed in these smaller studies. This potential limitation was accepted given the broader intent of this study was to inform the development of a COS for future prospective trials. The frequently reported outcomes may also not be relevant to key stakeholders, as demonstrated by a profound lack of patient-centred outcome measures.
One hundred and two discreet adverse event outcomes were reported, many of them only appearing in a small number of studies. This is likely a representation of papers investigating NSTIs affecting specific anatomical regions (e.g., craniofacial NSTI) and reporting anatomically specific outcomes (e.g., proptosis) that would not be generalisable or relevant to all studies of NSTI.
When comparing outcome measures reported by studies published between 2010–2015 to those published between 2016–2020 a possible trend towards reporting more specific outcomes is noted. Vague outcome measures such as ‘organ failure/dysfunction’ and ‘mortality without time specified’ became less frequent, whilst more specific outcomes such as 28-day mortality, 30-day mortality, 90-day mortality appear more frequently. This is consistent with an increased emphasis on reporting transparency through preregistration of study protocols, which aims to decrease the risk of data being manipulated to support a hypothesis.16 Also of note is that patient reported outcomes such as Medical Outcomes Short Form-36 (SF-36) and pain score (visual analogue scale) were reported more frequently in the latter period, however the total number of studies utilising these outcomes remains very low.
Although a trend towards more specific outcome reporting is promising, in the absence of a COS the ability to generalise data is still limited. As has been previously noted in the literature, the limited number of studies that have investigated HBOT and other adjuvant therapies for NSTI have not reported consistent outcome sets,7,17–19 posing significant challenges in performing meta-analyses.3 This is a particularly important issue in NSTI given the rarity of the condition as well as the paucity of high-quality prospective trials. Thus, there remains ongoing discourse regarding the role of HBOT and other measures in NSTI management. The inconsistency in reporting is evidenced in this review by mortality being reported in 23 different ways with varying time points or qualifiers.
Quality assessments of the included studies were not performed, as examination and synthesis of data was beyond the scope of this review. In developing a COS, it may be useful to further investigate the outcome measures utilised specifically in high quality studies. Potential weaknesses of this review include that the search was limited to English language results (although most studies identified and included were produced in countries where English is not the official language) and the exclusion of studies which reported solely laboratory-based outcome measures. Exclusion of qualitative outcomes that were neither pooled nor tabulated is another potential, although likely minor, limitation.
This study is the first in a series that aims to develop a COS for NSTI. It offers an inventory of outcomes reported in NSTI research which can now be proposed to an expert panel through a Delphi study, for determination of the most important outcomes to be included in future trials.
Conclusion
This systematic review provides a comprehensive inventory of the outcome measures currently being utilised for NSTI research and demonstrates a marked heterogeneity in outcome reporting. This inventory is a critical first step in the development of a COS, a process which is now underway in a separate Delphi study.
References
- Stevens D, Bryant A. necrotising soft-tissue infections. N Engl J Med. 2017;377:2253–65. doi: 10.1056/NEJMra1600673. PMID: 29211672.
Hedetoft M, Madsen M, Madsen L, Hyldegaard O. incidence, comorbidity and mortality in patients with necrotising soft-tissue infections, 2005–2018: a Danish nationwide register-based cohort study. BMJ Open. 2020;10:e041302. doi: 10.1136/bmjopen-2020-041302. PMID: 33067303. PMCID: PMC7569942. - Hedetoft M, Bennett M, Hyldegaard O. adjunctive hyperbaric oxygen treatment for nectorising soft-tissue infections: A systematic review and meta-analysis. Diving Hyperb Med. 2021;51:34–43. doi: 10.28920/dhm51.1.34-43. PMID: 33761549. PMCID PMC8081587.
- Williamson P, Altman D, Bagley H, Barnes K, Blazeby, J, Brookes S, et al. the COMET handbook: version 1.0. Trials. 2017;18:280. doi: 10.1186/s13063-017-1978-4. PMID: 28681701. PMCID: PM5499094.
- Boers M, Kirwan J, Wells G, Beaton D, Gossec L, d’Agostino M, et al. Developing core outcome measurement sets for clinical trials: OMERACT filter 2.0. J Clin Epidemiol. 2014;67:745–53. doi: 10.1016/j.jclinepi.2013.11.013. PMID: 24582946.
- COMET-Initiative. COMET Database. Liverpool, United Kingdom. [cited 2023 Apr 25]. Available from: https://www.comet-initiative.org/Studies.
- Hua C, Bosc R, Sbidian E, De Prost N, Hughes C, Jabre P, et al. Interventions for necrotizing soft tissue infections in adults. Cochrane Database Syst Rev. 2018;5:CD011680. doi: 10.1002/14651858.CD011680.pub2. PMID: 29851032. PMCID: PMC6494525.
- Page M, McKenzie J, Bossuyt P, Boutron I, Hoffman T, Mulrow C, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71. PMID: 33782057. PMCID: PMC8005924.
- Harris P, Taylor R, Thielke R, Payne J, Gonzalez N, Conde J. Research electronic data capture (REDCap) - a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–81. doi: 10.1016/j.jbi.2008.08.010. PMID: 18929686. PMCID: PMC2700030.
- Dodd S, Clarke M, Becker L, Mavergames C, Fish R, Williamson P. A taxonomy has been developed for outcomes in medical research to help improve knowledge discovery. J Clin Epidemiol. 2018;96:84–92. doi: 10.1016/j.jclinepi.2017.12.020. PMID: 29288712. PMCID: PMC 5854263.
- Clarivate Analytics. EndNote X8. Philadelphia, United States of America. [cited 2023 Apr 25]. Available from: https://endnote.com/downloads.
- Vertias Health Innovation. Covidence systematic review software. Melbourne, Australia. [cited 2023 Apr 25]. Available from: https://www.covidence.org/.
- Bulger E, May A, Dankner W, Maislin G, Robinson B, Shivran A. validation of a clinical trial composite endpoint for patients with necrotizing soft tissue infections. J Trauma Acute Care Surg. 2017;83:622–7. doi: 10.1097/TA.0000000000001564. PMID: 28538644.
- Williamson P, Altman D, Blazeby J, Clarke M, Gargon E. Driving up the quality and relevance of research through the use of agreed core outcomes. J Health Serv Res Policy. 2012;17:1–2. doi: 10.1258/jhsrp.2011.011131. PMID: 22294719.
- Nawin F, Smeeing D, Houwert R, Leenen L, Hietbrink F. Time is of the essence when treating necrotizing soft tissue infections: a systematic review and meta-analysis. World J Emerg Surg. 2020;15:4. doi: 10.1186/s13017-019-0289-6. PMID: 31921330. PMCID: PMC6950871.
- Mitra-Majumdar M, Kesselheim A. reporting bias in clinical trials: progress toward transparency and next steps. PLoS Med. 2022;19:e1003894. doi: 10.1371/journal.pmed.1003894. PMID: 35045078. PMCID: PMC8769309.
- Devaney B, Frawley G, Frawley L, Pilcher DV. Necrotising soft tissue infections: the effect of hyperbaric oxygen on mortality. Anaesth Intensive Care. 2015;43:685–92. doi: 10.1177/0310057X1504300604. PMID: 26603791.
- Soh CR, Pietrobon R, Freiberger JJ, Chew ST, Rajgor D, Gandhi M, et al. Hyperbaric oxygen therapy in necrotising soft tissue infections: a study of patients in the United States Nationwide Inpatient Sample. Intensive Care Med. 2012;38:1143–51. doi: 10.1007/s00134-012-2558-4. PMID: 22527074.
- Creta M, Longo N, Arcaniolo D, Giannella R, Cai T, Cicalese A, et al. Hyperbaric oxygen therapy reduces mortality in patients with Fournier’s Gangrene. Results from a multi-institutional observational study. Minerva Urol Nefrol. 2020;72:223–8. doi: 10.23736/S0393-2249.20.03696-6. PMID: 32083420.
Acknowledgements
The authors acknowledge Monash University for providing access to Covidence systematic review software and Alfred Health for providing access to REDCap.
Conflicts of interest and funding: nil
Submitted: 10 November 2023
Accepted after revision: 1 January 2024
Copyright: This article is the copyright of the authors who grant Diving and Hyperbaric Medicine a non-exclusive licence to publish the article in electronic and other forms
Appendix 1
A supporting document that outlines the search strategy employed in our OVID Medline search so that the search could be repeated and readers can better understand our search terms
Appendix 2
A supporting document that provides references for the studies ultimately included by our review for data extraction.
Appendix 3
A supporting document that lists all outcome measures and their relative frequency that were identified by the study, having been stratified into groups based on their content. This list provides readers with a comprehensive account of outcomes currently being reported in NSTI research should they need it for further work.
- Super User
- Category: Information
- 21547
2023 December;53(4)(Suppl)
Diving Hyperb Med. 2023 December 20;53(4Suppl):1−17. doi: 10.28920/dhm53.4.suppl.1-17. PMID: 38092370. PMCID: PMC10911829.
A European code of good practice for hyperbaric oxygen therapy - Review 2022
Jacek Kot, Secretary (Poland), Jordi Desola (Spain), Folke Lind (Sweden), Peter Mueller (Switzerland), Erik Jansen (Denmark), Francois Burman (South Africa)

Prepared by the Working Group «SAFETY»of the COST Action B14 «HYPERBARIC OXYGEN THERAPY»
May 2004 – Update 2022
Corresponding author: Prof. Jacek Kot, MD, PhD, ECHM President, National Centre for Hyperbaric Medicine, Institute of Maritime and Tropical Medicine in Gdynia, Medical University of Gdansk, Powstania Styczniowego 9B, 81-519 Gdynia, Poland
This version of the Code is an update of the first edition of the document (published May 2004). The original 2004 edition was prepared by the Working Group “SAFETY” of the COST Action B14 “HYPERBARIC OXYGEN THERAPY” by Jacek Kot, Secretary (Polland), Jordi Desola (Spain), Antonio Gata Simao (Portugal), Roly Gough- Allen (United Kingdom), Robert Houman (Belguim), Jean-Louis Meliet / Francois Galland (France), Christian Mortensen (Denmark), Peter Mueller (Denmark), Seppo Sipinen (Finland) and approved by the ECHM Board of Representatives. The current review was approved by the ECHM Board of Representatives and endorsed by the EUBS.
The following major modifications have been introduced in the current version:
- The new job of the Safety Manager has been added to the list of hyperbaric facility staff.
- Functions for hyperbaric facility staff have been described in the main text. The specific requirements for their background as well as for initial and continuous education has been left in external, referenced documents.
- The system for classification of hyperbaric chambers, hyperbaric systems and hyperbaric facilities has been introduced.
- The number of chambers, which can be operated simultaneously by one chamber operator, and the number of patients, which can attend inside the hyperbaric multiplace chamber, depending on the patients’ clinical status, have been added.
- Description of clinical indications and contraindications have been added to the main text.
- Three Annexes (1 - ECHM Educational and Training Standards for the Staff of Hyperbaric Medical Centres dated 1997; 2 – ECHM Recommendations for Safety in Multiplace Medical Hyperbaric Chambers dated 1998; 3 – on COST-B14 Working Group <Technical Aspects> Final Report dated 2001) have been removed from the main text (but referenced externally), with all their major conclusions being kept in the Code.
- The Risk Assessment for installation of the monoplace chamber has been added as an Annex 5. This Annex has been prepared by Francois Burman within his extensive and long-lasting cooperation with ECHM.
Copyright: This article is the copyright of the authors who grants Diving and Hyperbaric Medicine a non-exclusive licence to publish the article in electronic and other forms.
Publication Type: Journal Supplement
Full supplement available here.
- Super User
- Category: Information
- 8403
2024 March;54(1Suppl)
Diving Hyperb Med. 2024 March 31;54(1Suppl):1−53. doi: 10.28920/dhm54.1.suppl.1-53. PMID: 38537300. PMCID: PMC11168797.
Decompression illness: a comprehensive overview
Simon J Mitchell1,2,3
1 Department of Anaesthesiology, School of Medicine, University of Auckland, Auckland, New Zealand
2 Department of Anaesthesia, Auckland City Hospital, Auckland, New Zealand
3 Slark Hyperbaric Medicine Unit, North Shore Hospital, Auckland, New Zealand
Corresponding author: Professor Simon Mitchell, Department of Anaesthesiology, School of Medicine, University of Auckland, Private Bag 92019, Auckland 1142, New Zealand
ORCiD: 0000-0002-5149-6371
Keywords
Arterial gas embolism; Decompression sickness; Pathophysiology; Prevention; Pulmonary barotrauma; Symptoms; Treatment
Abstract
(Mitchell SJ. Decompression illness: a comprehensive overview. Diving and Hyperbaric Medicine. 2024 March 31;54(1Supp):1−53. doi: 10.28920/dhm54.1.suppl.1-53. PMID: 38537300. PMCID: PMC11168797.)
Decompression illness is a collective term for two maladies (decompression sickness [DCS] and arterial gas embolism [AGE]) that may arise during or after surfacing from compressed gas diving. Bubbles are the presumed primary vector of injury in both disorders, but the respective sources of bubbles are distinct. In DCS bubbles form primarily from inert gas that becomes dissolved in tissues over the course of a compressed gas dive. During and after ascent (‘decompression’), if the pressure of this dissolved gas exceeds ambient pressure small bubbles may form in the extravascular space or in tissue blood vessels, thereafter passing into the venous circulation. In AGE, if compressed gas is trapped in the lungs during ascent, pulmonary barotrauma may introduce bubbles directly into the pulmonary veins and thence to the systemic arterial circulation. In both settings, bubbles may provoke ischaemic, inflammatory, and mechanical injury to tissues and their associated microcirculation. While AGE typically presents with stroke-like manifestations referrable to cerebral involvement, DCS can affect many organs including the brain, spinal cord, inner ear, musculoskeletal tissue, cardiopulmonary system and skin, and potential symptoms are protean in both nature and severity. This comprehensive overview addresses the pathophysiology, manifestations, prevention and treatment of both disorders.
Copyright: This article is the copyright of the authors who grants Diving and Hyperbaric Medicine a non-exclusive licence to publish the article in electronic and other forms.
Publication Type: Journal article
Full article available here.
- Super User
- Category: Information
- 333